For most of the history of mining, pyrite has been the mineral that breaks your heart. Forty-niners panned it out of California streams thinking they had struck it rich. Sixteenth-century Spanish prospectors dragged it across the Atlantic by the cartload. The brassy cubes glitter exactly like gold, and they are worth almost nothing. The whole reason pyrite got the nickname "fool's gold" is that for five hundred years it has been the booby prize of the mineral world, a crystal that seems to promise something it cannot deliver.
That is no longer quite true. In a study released through ScienceDaily and Eureka Alert this spring, a research team at West Virginia University reported that pyrite from Appalachian black shale contains measurable, recoverable lithium, the metal that powers every electric vehicle and grid-scale battery now being installed across the country. The amounts are not trivial. Their best samples liberated up to 54 percent of their total lithium load from the pyrite alone. If the pattern holds beyond the study site, the rock that has disappointed prospectors since the Renaissance may quietly become one of the more interesting energy materials in North America.
What "Fool's Gold" Actually Is, and Why Nobody Looked Inside It
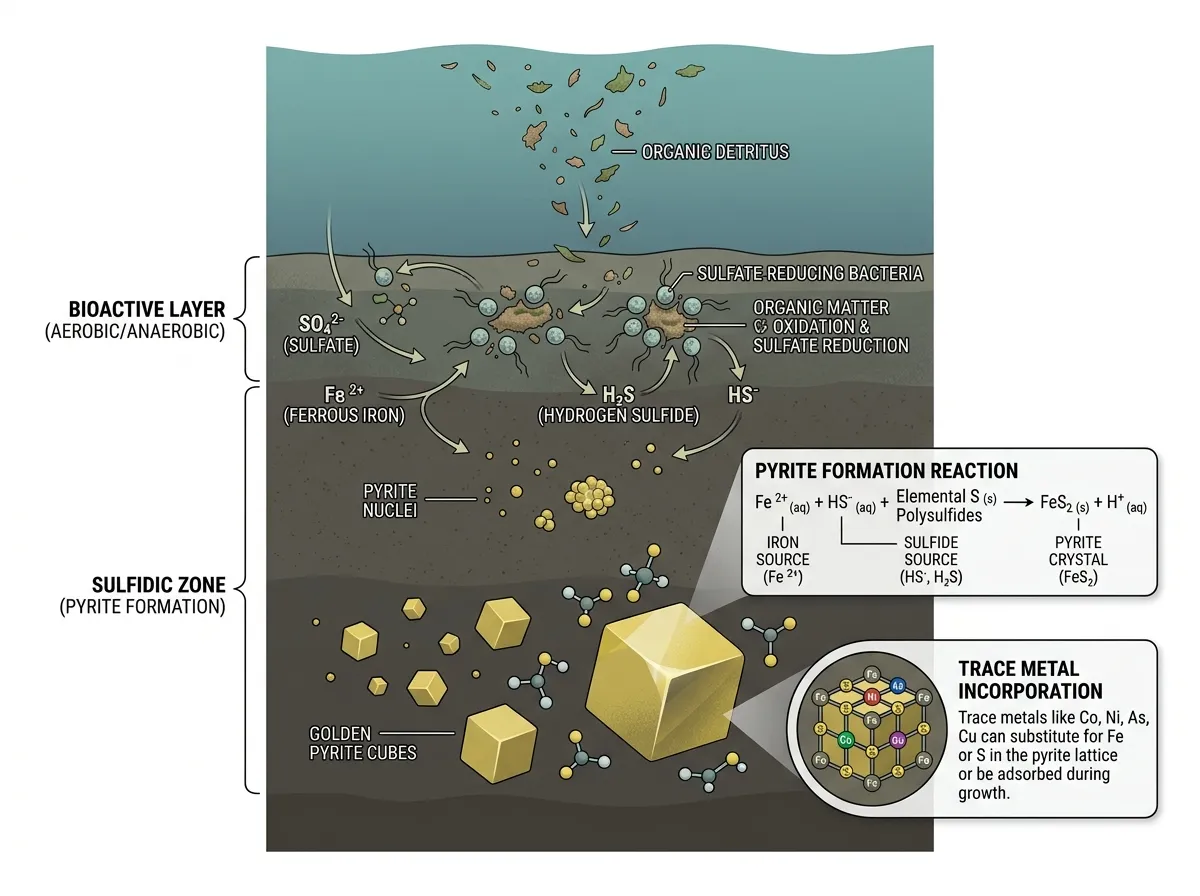
Pyrite is iron disulfide, FeS₂, a cubic crystal that forms in low-oxygen environments wherever iron and sulfur happen to meet. It grows in seafloor mud, in coal seams, in hot hydrothermal veins, and in the organic sludge that becomes black shale millions of years later. It is one of the most common sulfide minerals on Earth, and for industrial purposes it has been useful mostly as a source of sulfuric acid. Roast it, capture the sulfur dioxide, and you have the chemical that runs fertilizer plants and lead-acid batteries. The iron has historically been treated as a byproduct, and any other elements riding along inside the crystal have been treated as contamination rather than value.
That assumption made some sense. Pyrite has always been understood as a "trace metal sponge." Geologists have known for decades that it can incorporate small amounts of cobalt, nickel, arsenic, gold, and other elements during its formation, and ore deposits are sometimes evaluated by what is hiding inside the pyrite alongside more obvious targets. But lithium has not been on that list. Lithium is a small, mobile cation that prefers to sit in clay minerals, in evaporite brines, or in pegmatite ores like spodumene. Mineralogy textbooks do not generally describe pyrite as a host for it. There was no obvious reason to look.
The West Virginia team had a different question in mind. Shailee Bhattacharya, a doctoral student working with Professor Shikha Sharma in the IsoBioGeM Lab, was studying whether industrial waste from oil and gas operations, drill cuttings, mine tailings, and the leftover rock that gets pulled out of Appalachian wells, might contain lithium that could be extracted without sinking a new mine. The Marcellus and Devonian shales of Appalachia are organically rich, full of the kind of fine-grained black mudstone that traps trace metals as it lithifies. If the lithium signal in those rocks was concentrated anywhere in particular, that was what the team wanted to know.

The Surprise Hiding in Appalachian Black Shale
The team analyzed fifteen Devonian shale samples of varying organic richness from wells drilled into the Appalachian basin. They separated the pyrite from the rest of the rock matrix and tested each fraction for lithium content. What they found, and what surprised them, was that the lithium was not spread evenly. It was concentrated, sometimes dramatically, inside the pyrite grains themselves.
Bhattacharya, in interviews coordinated through West Virginia University communications, described the result in precise terms. The team detected significant lithium inside pyrite, "which is unheard of." In samples with relatively modest total lithium loads of around 22 parts per million, more than half of that lithium, up to 54 percent, came out of the pyrite fraction alone. The team also found a positive correlation between how much pyrite a sample contained and how much lithium they could recover from it, which is the kind of clean linear relationship that signals a real geochemical association rather than measurement noise.
The numbers need context. Twenty-two parts per million is not a brine deposit and it is not a hard-rock spodumene ore. The famous lithium brines of the Salar de Atacama in Chile run hundreds to thousands of parts per million in the pumped fluid. Australian spodumene mines yield ore that is one to two percent lithium oxide by weight. Black shale will not compete with either of those on grade. What it has instead is volume and access. The Marcellus and Utica shales already have thousands of producing wells, decades of accumulated drill cuttings, and an entire industrial logistics chain pointed at them. The lithium does not have to be rich. It has to be reachable.
How Lithium Got Trapped in Pyrite in the First Place
The mechanism the team is now trying to nail down has to do with how pyrite grows in low-oxygen marine mud. When organic matter rains down through a stratified water column and hits a seafloor where bacteria are reducing sulfate to sulfide, iron sulfide minerals begin to form in the sediment pore water. They start as soft, framboidal aggregates and recrystallize over time into the harder cubic pyrite that survives into the rock record. While they grow, they scavenge whatever ions are floating in that pore water, particularly elements that bind tightly to sulfide.
Lithium is not, on paper, a sulfide-loving element. It does not sit happily in a sulfide lattice the way zinc or cadmium does. The current working hypothesis is that lithium is not substituting cleanly for iron in the pyrite crystal structure. Instead, it is probably adsorbed onto pyrite surfaces during the growth phase, or trapped in tiny inclusions of clay and organic matter that pyrite engulfs as it expands. The recrystallization that hardens framboidal pyrite into euhedral crystals can lock those impurities in place permanently. By the time the rock is sitting in an Appalachian outcrop 380 million years later, the lithium is no longer mobile in groundwater. It is structurally bound to a mineral that nobody thought to look at.
If that mechanism turns out to be correct, it has implications beyond Appalachia. Black shales rich in pyrite are present on every continent. The Posidonia Shale of Germany, the Bazhenov Formation of western Siberia, the Vaca Muerta of Argentina, and the Bakken of North Dakota are all candidates for the same kind of analysis. None of these formations were on lithium maps before. They could be now.
The Mining-Waste Math Nobody Has Done
The most interesting feature of the discovery is not the geology. It is what happens when you put the numbers next to existing waste streams. The Appalachian basin currently produces drill cuttings on a staggering scale. A single horizontal Marcellus well generates roughly two thousand cubic feet of cuttings, and there are something like fifteen thousand active wells across the play. Cuttings are typically disposed of in lined landfills or used as low-grade fill material. They are, for now, an environmental liability rather than an asset.
Run the back-of-envelope arithmetic. If even a small fraction of those cuttings carries pyrite at the modest concentrations Bhattacharya's group reported, and if the recovery efficiency holds up at industrial scale, the United States may already be sitting on a meaningful domestic lithium reserve that nobody has assayed. The country imports more than ninety percent of its lithium today, mostly from Australia and Chile, with a growing share processed through Chinese refining capacity. Domestic production has been concentrated at a single Nevada brine operation, with a much-discussed second source potentially coming from the McDermitt Caldera on the Nevada-Oregon border. Adding Appalachian black shale to that map would change the geography of American lithium in a way no policy document has yet contemplated.
There are real technical hurdles. The leaching processes Bhattacharya's group used in the lab are not yet scaled-up industrial protocols. Recovering lithium from pyrite at sub-economic grades will require either very low extraction costs or a co-product strategy that monetizes the iron and sulfur as well. The acid produced by oxidizing pyrite is also a notorious environmental hazard. Acid mine drainage from old pyrite-rich coal mines is one of the most persistent water quality problems in Appalachia, and any large-scale program that handled pyrite waste would need a closed-loop system that does not make that problem worse. None of these are dealbreakers. They are, instead, the engineering questions that turn an interesting laboratory finding into a real industry.
Why This Matters Beyond Batteries
There is a broader pattern here that geochemists have been quietly documenting for two decades. The "ore deposit" framework that organized twentieth-century mining, which assumes that economically useful elements are concentrated in specific, identifiable, mineable bodies, is increasingly being supplemented by a different model. In the new model, those same elements are dispersed at low concentrations across enormous volumes of rock and waste, and the engineering challenge is not to find them but to extract them efficiently from material that is already on the surface. Rare earth elements in coal fly ash, scandium in red mud from aluminum production, cobalt and nickel in deep-sea polymetallic nodules, and now lithium in shale pyrite, are all examples of the same shift.
The shift matters because it changes the political economy of critical minerals. A traditional mine is a fixed point on a map, controlled by whoever owns the land and the permits. A waste stream is everywhere, and it is owned by whoever generated it. If the lithium economy starts pulling material out of existing drill cuttings, fly ash piles, and acid mine drainage ponds, the supply geography of the metal will look very different from the supply geography of the ores it currently displaces. Countries with long industrial histories and large legacy waste inventories, the United States very much included, may end up with strategic positions they did not realize they had.
It is also a useful corrective to a kind of resource pessimism that has crept into the energy transition conversation. The argument that "we cannot mine our way to a clean energy future" has rhetorical force, and there are real ecological reasons to take it seriously. But it tends to assume that the resources available are the resources we currently know about. The history of mineralogy is full of moments where someone looked carefully at a rock that everyone had ignored and found something useful inside it. Pyrite happens to have been ignored for a particularly long time.
The Caveats Researchers Want You to Know
Bhattacharya and Sharma have been careful in their own communications to say what the study does not show. The samples come from a single study site in the Appalachian basin. The researchers note that the observations cannot be extrapolated beyond those samples without further work. Whether the same relationship holds across other shale formations, other geological ages, and other tectonic settings is a question for the next round of fieldwork, not for this paper.
The leaching efficiencies are also lab-bench numbers, run on small, hand-prepared samples under controlled conditions. Industrial extraction has to deal with mixed feedstock, water chemistry, energy costs, and waste handling at scales that do not appear in a published paper. There is also a real possibility, which the team flags, that lithium concentrations vary substantially within a single formation depending on local depositional conditions. Some Appalachian wells will turn out to be much richer than others, and predicting which is which is going to require the kind of geochemical mapping that has not yet been done.
That uncertainty is normal for an early-stage discovery. What is striking is that the basic claim, that pyrite hosts measurable lithium in organic-rich black shale, was missing from the literature until very recently and is now being supported by an independent line of evidence. Mining companies, including some of the larger Appalachian gas operators, have started commissioning their own assays of cuttings and waste rock. The next two years of follow-up studies will determine whether this becomes a footnote in a textbook or a line item on a federal critical-minerals strategy.

Where This Could Lead
The pyrite story is, in the end, a story about looking again. Geologists have been holding samples of black shale in their hands for over a century. They have been looking at pyrite cubes through microscopes for longer than that. The lithium was there the whole time. It took a specific question, asked by a doctoral student at a regional university working on an ostensibly applied problem, for someone to actually measure it.
That has happened before, often enough that it should not surprise anyone. The fertilizer revolution started with someone wondering what was in bird guano. The semiconductor industry started with someone noticing odd electrical behavior in samples of impure germanium. The pattern is consistent. The world is full of materials that have been quietly mislabeled as worthless because nobody asked the right question. Fool's gold has been the textbook example of a worthless mineral for five centuries. It now turns out, in at least some samples, to contain the metal that runs every laptop, every phone, every electric car, and every grid battery being installed in 2026. The next question is which other rocks have been waiting for the same conversation.
Sources
- Fool's gold isn't so foolish: Scientists find hidden treasure in pyrite, ScienceDaily
- Pyrite, also known as fool's gold, may contain valuable lithium, a key element for green energy, EurekAlert
- Researchers point to pyrite as possible source of lithium, MINING.COM
- Discovery in fool's gold: Shale pyrites hold hidden lithium riches, Interesting Engineering
- Potential lithium enrichment in pyrites from organic-rich shales, NASA ADS

