The gas that keeps you alive nearly wiped out all life on Earth. That sentence is not a paradox. It is a summary of one of the most consequential events in the planet's 4.5-billion-year history, and understanding how it happened requires thinking about chemistry, climate, and evolution on timescales that dwarf anything in human experience.
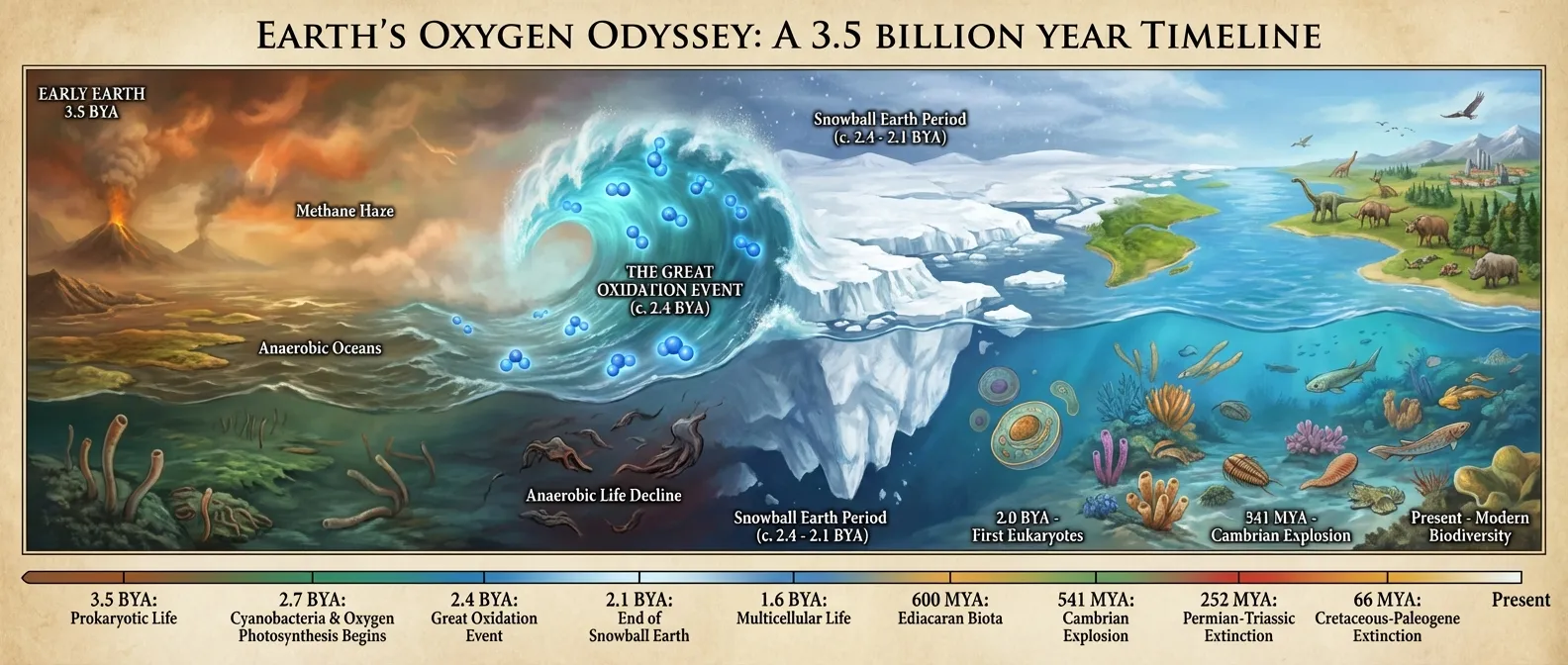
Roughly 2.4 billion years ago, a group of photosynthetic microorganisms called cyanobacteria figured out how to split water molecules using sunlight. The energy payoff was enormous. The waste product was molecular oxygen. And the world that oxygen entered was completely unprepared for it. What followed, known as the Great Oxidation Event (sometimes called the Oxygen Catastrophe), set off a chain of consequences that reshaped the atmosphere, froze the planet, and eventually made complex life possible. It is a story about innovation, catastrophe, and the strange way that destruction can become the foundation for everything that comes after.
A World Without Oxygen
To understand what happened, you first need to understand the world that oxygen entered. Early Earth's atmosphere was dominated by nitrogen, carbon dioxide, methane, and water vapor. There was essentially no free oxygen. The oceans were rich in dissolved iron, giving them a greenish tint, and the chemistry of life was built entirely around anaerobic metabolism: extracting energy from chemical reactions that don't involve oxygen.
This wasn't a primitive arrangement waiting to be improved. Anaerobic life was extraordinarily successful. For roughly 1.5 billion years before the Great Oxidation Event, microbial communities flourished in a world where oxygen would have been as toxic to them as cyanide is to us. They had colonized every available niche in the oceans and built complex ecosystems based on sulfur chemistry, iron chemistry, and methanogenesis (the production of methane). Methanogens, in particular, were critical to Earth's climate: the methane they produced acted as a powerful greenhouse gas, keeping the planet warm despite a sun that was roughly 20 to 25 percent dimmer than it is today.
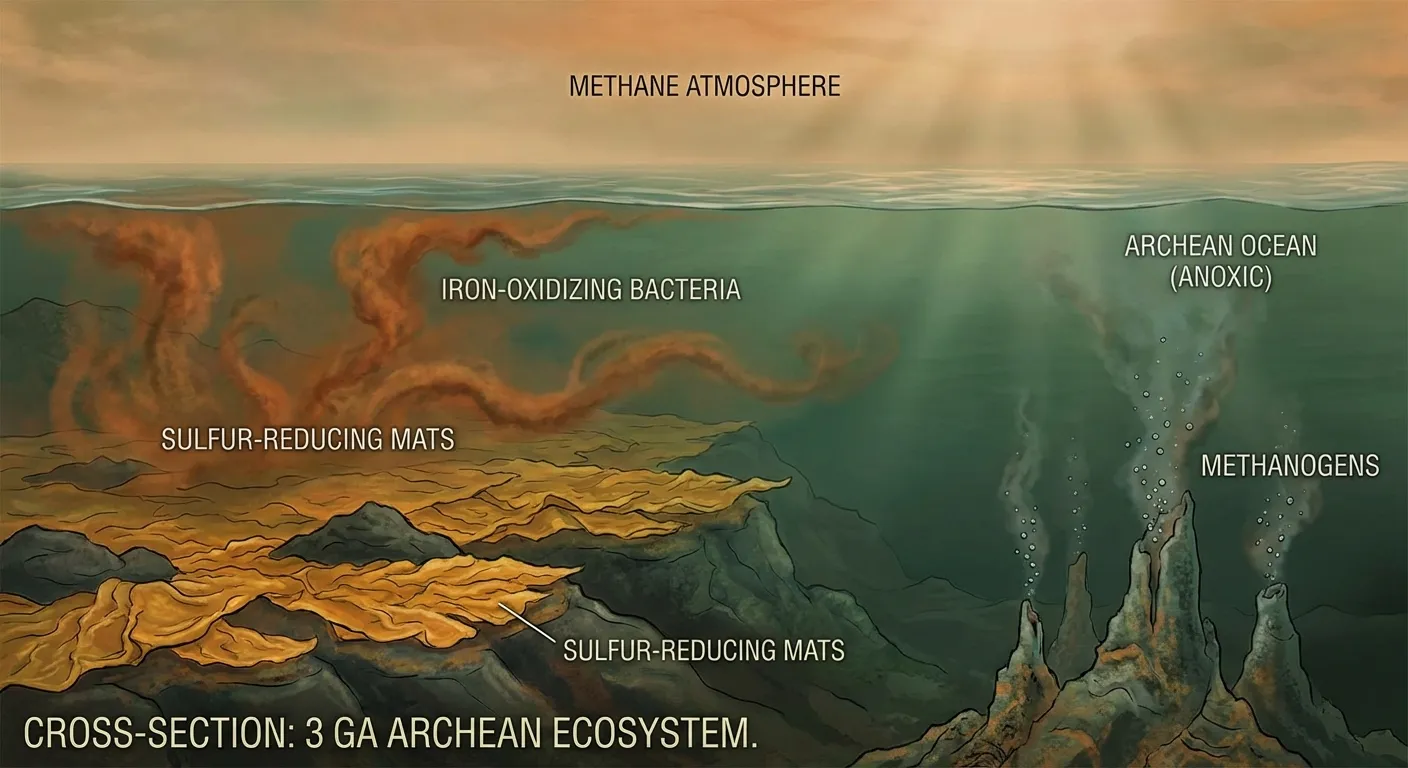
Into this stable, methane-warmed, oxygen-free world came the cyanobacteria, and their invention of oxygenic photosynthesis changed everything.
The Invention That Changed Everything
Photosynthesis itself wasn't new. Earlier microorganisms had evolved forms of photosynthesis that used hydrogen sulfide or iron as electron donors, producing sulfur or rust as byproducts. These reactions were relatively low-energy. What cyanobacteria invented was fundamentally different: they learned to use water as an electron donor, a far more abundant resource, but one that required much more energy to split. The payoff was enormous. The byproduct was molecular oxygen, O2.
The exact timing of this invention is debated. Molecular evidence suggests cyanobacteria capable of oxygenic photosynthesis may have evolved as early as 3 billion years ago, roughly 600 million years before the Great Oxidation Event itself. If that timeline is correct, it raises a question: why did it take so long for oxygen to accumulate in the atmosphere?
The answer involves what geochemists call oxygen sinks. For hundreds of millions of years, every molecule of oxygen that cyanobacteria produced was immediately consumed by chemical reactions with dissolved iron in the oceans and with volcanic gases in the atmosphere. The oxygen reacted with iron to form iron oxides, which precipitated out of the water and settled on the ocean floor as banded iron formations, the distinctive red-and-gray striped rocks that are now mined as iron ore across the world. These formations are, in a sense, the fossil record of oxygen's slow war of attrition against a planet that was chemically hostile to it.
Eventually, the sinks were overwhelmed. The dissolved iron was used up. The volcanic gases couldn't absorb oxygen fast enough. And sometime around 2.4 billion years ago, free oxygen began accumulating in the atmosphere for the first time. The concentration was still tiny by modern standards, perhaps 1 to 2 percent of present levels, but for organisms that had never encountered it, even trace amounts were lethal.
The Oxygen Catastrophe
Oxygen is, from a biochemical standpoint, a dangerous molecule. It is highly reactive, eagerly stripping electrons from other compounds in a process called oxidation. This is the same chemistry that rusts iron, that causes a cut apple to turn brown, and that makes fire possible. For organisms whose metabolic machinery had evolved in the absence of oxygen, this reactivity was devastating. Oxygen attacked their enzymes, damaged their DNA, and disrupted the chemical reactions they depended on for energy.
The result was a mass extinction that, in terms of the proportion of species eliminated, may have been the most severe in Earth's history. The vast majority of anaerobic life was killed outright or driven into the shrinking number of environments where oxygen couldn't reach: deep ocean sediments, mud at the bottom of lakes, the guts of other organisms. These are the same niches where obligate anaerobes survive today, billions of years later, refugees from a catastrophe that reshaped the planet.

But the catastrophe didn't stop at biology. Oxygen's arrival triggered a cascade of geochemical changes that nearly froze Earth solid. The methane that had been keeping the planet warm was itself vulnerable to oxidation. As atmospheric oxygen reacted with methane, it converted this potent greenhouse gas into carbon dioxide and water, both of which are far weaker at trapping heat. With the methane greenhouse collapsing and the sun still 20 percent dimmer than today, global temperatures plummeted.
The Snowball
What followed was one of the most extreme climate events in Earth's history: the Huronian glaciation, also known as the Paleoproterozoic Snowball Earth. Ice sheets advanced from the poles toward the equator, and geological evidence suggests that ice may have covered the entire planet, or very nearly so, for tens of millions of years.
A 2020 study published in Proceedings of the National Academy of Sciences confirmed that the Great Oxidation Event preceded the Paleoproterozoic Snowball Earth glaciation, establishing the causal chain: cyanobacteria produced oxygen, oxygen destroyed atmospheric methane, methane loss collapsed the greenhouse effect, and global temperatures crashed. The study dated the GOE to approximately 2,501 to 2,434 million years ago, placing it squarely before the glacial deposits found in South Africa and Russia.
The Snowball Earth phase was, in some respects, as severe as the oxygen poisoning itself. Photosynthesis would have been dramatically curtailed under global ice cover, and the cyanobacteria that had triggered the crisis would have been among its victims. Life persisted in refugia: around volcanic hot springs, in pockets of liquid water beneath the ice, in the thin band of open ocean that may have survived near the equator. But the biosphere was reduced to a shadow of its former self, squeezed between the twin catastrophes of oxygen toxicity and global freezing.
Creative Destruction
The Snowball eventually ended. Volcanic CO2 accumulated in the atmosphere over millions of years (since there was no liquid water to absorb it through weathering), eventually building a greenhouse effect strong enough to melt the ice. When the thaw came, it came fast, and the world that emerged was radically different from the one that had frozen.
Oxygen was now a permanent feature of the atmosphere. And while it had been lethal to anaerobic life, it offered a staggering metabolic advantage to any organism that could learn to use it. Aerobic respiration, the process of extracting energy from organic molecules using oxygen, is approximately 18 times more efficient than anaerobic metabolism. An organism that could tolerate and exploit oxygen had access to an energy source that its anaerobic competitors simply couldn't match.
This energy dividend was the engine of biological complexity. The reason you have a brain, muscles, and organs that consume enormous amounts of energy is that aerobic metabolism makes such energy-hungry systems viable. Without the oxygen that cyanobacteria pumped into the atmosphere 2.4 billion years ago, complex multicellular life, from the earliest animals to every species living today, would be energetically impossible.
The parallel to disruptive technologies is hard to ignore. Cyanobacteria didn't intend to reshape the planet. They were simply exploiting an abundant resource (water) with a novel biochemical technique (oxygenic photosynthesis), and the waste product of their success (oxygen) destroyed the existing order. The deep ocean ecosystems that thrive in darkness today are, in a sense, remnants of the pre-oxygen world: communities built on chemosynthesis rather than photosynthesis, surviving in the few environments where the ancient anaerobic chemistry still works.
Life as Its Own Worst Enemy
The Great Oxidation Event is sometimes framed as a triumph: life invented photosynthesis, oxygenated the atmosphere, and paved the way for complex organisms. That framing is accurate but incomplete. It treats the cyanobacteria as liberators when they were, more precisely, the most successful invasive species the planet has ever produced. They did not set out to transform the world. They exploited an abundant resource, and the waste product of their success happened to be incompatible with the biochemistry of nearly everything else alive.
This pattern, where life's innovations create environmental crises that in turn drive new evolutionary opportunities, has repeated throughout Earth's history. The Cambrian Explosion, the colonization of land, the evolution of grass-dominated ecosystems, each of these transitions involved organisms that fundamentally altered their environment, often at enormous cost to existing species, and opened new ecological possibilities that didn't exist before.
Understanding the Great Oxidation Event matters beyond geology because it illuminates a fundamental dynamic of complex systems. Innovation and destruction are not opposites. They are, at the deepest level, the same process viewed from different timescales. The ongoing discovery of new species in previously unexplored environments is a reminder that the biosphere is still shaped by the chemical revolution that began 2.4 billion years ago, when a microorganism's metabolic waste product became the most important molecule in the history of life.
Why It Matters
Every breath you take is a consequence of the Great Oxidation Event. The 21 percent oxygen in the air around you traces directly back to the cyanobacterial innovation that reshaped this planet's chemistry. The iron in your blood, carried by hemoglobin to fuel your cells, exists in its current oxidized form because of what those microorganisms set in motion billions of years ago. Even the ozone layer that shields the surface from ultraviolet radiation is a downstream product of atmospheric oxygen.
The event also carries a cautionary resonance, though not the one usually cited. The standard analogy compares cyanobacteria to humans: both species altered the atmosphere with a waste product. But the deeper lesson may be about timescale. The Great Oxidation Event took hundreds of millions of years from the evolution of oxygenic photosynthesis to the atmospheric tipping point. The consequences unfolded over hundreds of millions more. The cyanobacteria had no capacity to notice what they were doing, let alone respond. Whether a species that can measure its own atmospheric changes in real time will prove more adaptive than one that could not is the question that makes this ancient story feel uncomfortably current.
Sources
- The Great Oxidation Event: How Cyanobacteria Changed Life (American Society for Microbiology)
- The Great Oxygenation Event: The Earth's First Mass Extinction (Slate)
- The Great Oxidation Event preceded a Paleoproterozoic "Snowball Earth" (PNAS, 2020)
- The Great Oxygenation Event as a consequence of ecological dynamics modulated by planetary change (Nature Communications, 2021)
- Billions of years ago, the rise of oxygen caused a worldwide deep freeze (The Conversation)

