Semaglutide is the active ingredient in Ozempic, Wegovy, and Rybelsus, three brand-name medications that have become the most talked-about drugs since the COVID vaccines. They're all the same molecule made by the same company (Novo Nordisk), but prescribed for different conditions at different doses. If you're confused about which is which and how any of them actually work in your body, you're in good company.
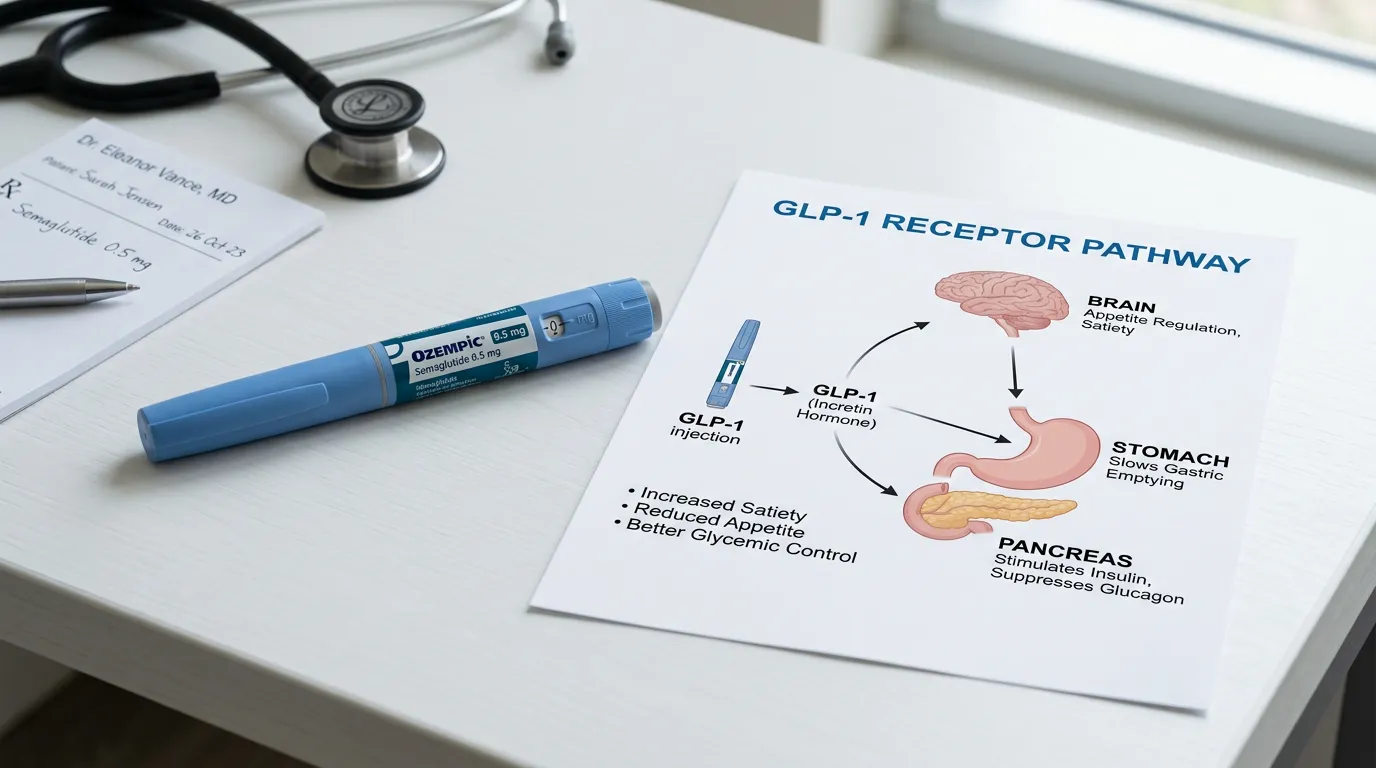
Here's the direct answer: semaglutide is a GLP-1 receptor agonist, which means it mimics a hormone your gut naturally produces after eating. That hormone tells your brain you're full, tells your pancreas to release insulin, and slows your digestion. The medication amplifies those signals, leading to reduced appetite, lower blood sugar, and, in most people, significant weight loss. The clinical data shows average weight loss of 15% of body weight over about a year, with some patients losing over 20%.
How Semaglutide Works in Your Body
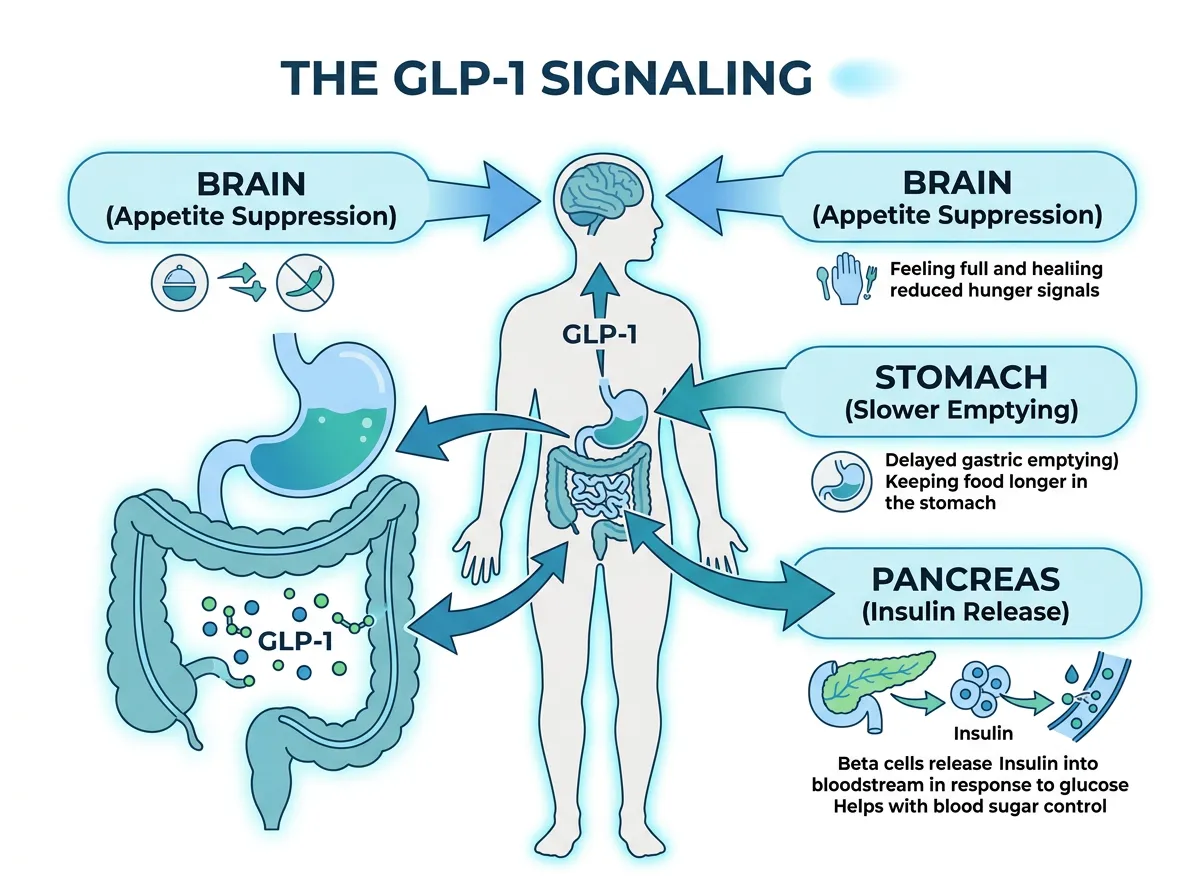
Your body naturally produces a hormone called GLP-1 (glucagon-like peptide-1) in your intestines when you eat. GLP-1 does several things simultaneously: it signals your pancreas to release insulin (which lowers blood sugar), it suppresses glucagon (a hormone that raises blood sugar), it slows the rate at which your stomach empties food into your intestines, and it signals your brain that you're satisfied. The problem is that natural GLP-1 breaks down in your bloodstream within minutes. Its effects are real but brief.
Semaglutide is an engineered version of GLP-1 that lasts much longer, roughly a week in injectable form. Harvard Health explains the mechanism clearly: it binds to the same receptors as natural GLP-1 but resists the enzymes that normally break the hormone down. This means a single weekly injection maintains elevated GLP-1 signaling continuously, producing sustained effects on appetite, blood sugar, and digestion that natural GLP-1 could never achieve.
In the brain, semaglutide targets the hypothalamus, specifically activating neurons that suppress appetite (POMC/CART neurons) while inhibiting neurons that stimulate hunger (NPY/AgRP neurons). This is why people on semaglutide describe a fundamental shift in their relationship with food: they stop thinking about it constantly, portion sizes that previously seemed small now feel adequate, and cravings, particularly for high-calorie foods, diminish substantially. It also appears to interact with dopamine reward pathways, which may explain emerging research into its potential effects on addictive behaviors beyond food.
A December 2024 review in *Current Issues in Molecular Biology* mapped out additional mechanisms: semaglutide promotes the conversion of white fat cells to brown fat cells through AMPK/SIRT1 activation, increasing the body's passive calorie burn, and reduces inflammatory markers including IL-6 and TNF-alpha. However, the review noted an important caveat: "most supporting evidence on mechanistic actions of semaglutide is preclinical, demonstrated in rodents and not actually confirmed in humans." The appetite and blood sugar effects are well-documented in people. The fat-cell conversion and anti-inflammatory mechanisms need more human research.
Ozempic vs. Wegovy vs. Rybelsus: Same Drug, Different Uses
All three are semaglutide. The difference is the approved condition, the dose, and the delivery method.
Ozempic is a once-weekly injection approved by the FDA for type 2 diabetes management. It starts at 0.25 mg per week and can be titrated up to 2 mg. In January 2025, the FDA expanded Ozempic's approval to include reducing the risk of kidney disease progression and cardiovascular death in adults with type 2 diabetes and chronic kidney disease. Ozempic is the version your doctor prescribes if you have type 2 diabetes. It's also the version most commonly prescribed off-label for weight loss, because Wegovy has been in chronic shortage, though using Ozempic specifically for weight loss is technically not an FDA-approved use.
Wegovy is a once-weekly injection approved specifically for chronic weight management. It uses a higher maximum dose (2.4 mg vs. Ozempic's 2 mg) and is indicated for adults with a BMI of 30 or higher, or 27 or higher with at least one weight-related health condition like hypertension, type 2 diabetes, or high cholesterol. In December 2025, the FDA approved an oral version of Wegovy as well, a 25 mg daily tablet for the same weight management indication.
Rybelsus is a daily oral tablet approved for type 2 diabetes, with a maximum dose of 14 mg. It was the first oral GLP-1 medication, and in October 2025 its FDA approval expanded to include reducing cardiovascular risk in adults with type 2 diabetes. Rybelsus must be taken on an empty stomach with only a small sip of water, and you can't eat or take other medications for 30 minutes afterward. This strict dosing requirement makes it less convenient than the weekly injection for many patients.
You cannot take more than one semaglutide product simultaneously. If your doctor switches you from Ozempic to Wegovy, or vice versa, you stop one before starting the other.
What the Clinical Trials Show
The evidence base for semaglutide is unusually strong by pharmaceutical standards. The STEP trial program (Semaglutide Treatment Effect in People with obesity) ran multiple large, randomized, placebo-controlled trials testing the weight management dose.
STEP 1, published in the New England Journal of Medicine with over 1,900 participants, found that patients taking semaglutide 2.4 mg weekly lost an average of 14.9% of their body weight over 68 weeks, compared to 2.4% in the placebo group. Across the full STEP program (trials 1, 3, 4, and 8), mean weight losses ranged from 14.9% to 17.4%. Real-world data from 2025, tracking patients outside of clinical trial conditions, confirmed a 15% average weight reduction at 12 months.
The oral form of Wegovy (25 mg daily) showed comparable results: 16.6% mean weight loss at 64 weeks with full adherence, with one-third of adherent participants achieving at least 20% weight loss versus under 3% with placebo.
For diabetes management, the PIONEER trials showed that oral semaglutide (Rybelsus 14 mg) reduced A1C by 1.1% versus 0.1% with placebo, and 64% of patients reached the target A1C of 7.0% compared to 21% on placebo. The cardiovascular data is equally notable: the SELECT trial showed a 20% reduction in major cardiovascular events in overweight and obese adults without diabetes, and the FLOW trial demonstrated a 24% reduction in the risk of kidney disease progression.
These aren't marginal effects. A 15% body weight loss means a 200-pound person losing 30 pounds over a year, and the cardiovascular and kidney benefits extend the medication's impact well beyond the scale.

Side Effects: What the Latest Data Shows
A joint Harvard and CDC study published in Annals of Internal Medicine in April 2025 analyzed emergency department visits among semaglutide users in 2022 and 2023. The findings were reassuring overall: fewer than 4 ED visits occurred per 1,000 semaglutide patients. Of those visits, roughly 70% involved gastrointestinal complaints (nausea, vomiting, abdominal pain, diarrhea, constipation), about 6% were allergic reactions, and approximately 17% were for hypoglycemia.
The hypoglycemia finding raised some eyebrows. Dr. Pieter Cohen, the study's senior author and an associate professor at Harvard Medical School, noted: "Those patients were taking semaglutide by itself... we'll need more research to see if semaglutide can rarely cause severe hypoglycemia." He emphasized that users must "eat regularly, even though they may not feel hungry," because the appetite-suppressing effect can lead people to skip meals, dropping blood sugar below safe levels.
The gastrointestinal side effects are the most common complaint and the primary reason people stop taking the medication. Nausea tends to be worst during dose escalation (when your dose is being gradually increased over the first 16 to 20 weeks) and typically improves as your body adjusts. Dr. Chika Anekwe at Harvard noted that "while gastrointestinal side effects like nausea and constipation are common, they are usually temporary and manageable."
More serious but rarer concerns include intestinal obstruction (the FDA added a safety warning to Ozempic's label about bowel obstruction), a possible small increase in risk of a rare eye condition called non-arteritic anterior ischemic optic neuropathy (NAION), and potential loss of lean muscle mass alongside fat loss. The muscle loss issue is particularly relevant for older adults and people who aren't exercising during treatment, as losing muscle while losing fat can create long-term metabolic and mobility problems.
Semaglutide caused thyroid C-cell tumors in rodent studies at clinically relevant doses. This risk has not been confirmed in humans, but it's why the medication carries a boxed warning against use in people with a personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2.
Cost and Who Can't Take It
Semaglutide remains expensive. Without insurance, Ozempic costs approximately $935 to $1,349 per month. With insurance coverage for type 2 diabetes, copays typically range from $25 to $150 monthly. Novo Nordisk offers a savings program for commercially insured patients: as low as $25/month with a maximum savings of $100/month for up to 48 months. New self-pay patients can access the lowest doses at $199/month for the first two months, rising to $349/month afterward.
Insurance coverage for weight management (Wegovy) is improving but inconsistent. About 49% of employer plans with 500 or more employees covered GLP-1 medications for weight loss in 2025. Medicare will not cover GLP-1s for weight loss until at least July 2026. If your insurance covers the diabetes indication but not weight management, your doctor may prescribe Ozempic for its approved use if you also have type 2 diabetes, which provides the weight loss benefit within the approved indication.
Semaglutide is contraindicated (should not be used) in people with a personal or family history of medullary thyroid carcinoma, Multiple Endocrine Neoplasia syndrome type 2, known hypersensitivity to semaglutide, or a history of pancreatitis (relative contraindication requiring careful monitoring). Pregnant or breastfeeding women should not take semaglutide and should stop the medication at least two months before a planned pregnancy. People with diabetic retinopathy, gallbladder disease, or gastroparesis should use the medication with caution and close monitoring.
What to Remember
Semaglutide (sold as Ozempic, Wegovy, and Rybelsus) works by mimicking the GLP-1 hormone that controls appetite, blood sugar, and digestion. Clinical trials show average weight loss of 15% over a year, significant A1C reduction in diabetes patients, and measurable cardiovascular and kidney benefits. Side effects are mostly gastrointestinal, typically temporary, and serious complications are rare.
The medication is not a standalone solution. It works best alongside dietary changes and exercise, and weight regain after stopping the medication is common if lifestyle habits haven't changed. The cost remains a barrier for many people, though insurance coverage and manufacturer programs have expanded. If you're considering semaglutide, talk to your doctor about which formulation fits your medical situation, whether your insurance covers it, and what a realistic treatment plan looks like for your goals.
Sources
- How Does Ozempic Work? Understanding GLP-1s - Harvard Health, April 2025
- Serious Side Effects From Semaglutide Are Uncommon - Harvard Health / CDC, April 2025
- Spotlight on the Mechanism of Action of Semaglutide - Current Issues in Molecular Biology, December 2024
- Once-Weekly Semaglutide in Adults with Overweight or Obesity (STEP 1) - New England Journal of Medicine
- Semaglutide (Subcutaneous Route) - Mayo Clinic