You've never heard of CAG-170. Neither had most microbiologists until a few weeks ago. That's because this group of gut bacteria has been hiding in plain sight, detectable only through its DNA, impossible to grow in a lab dish, and yet present in the guts of healthy people on every inhabited continent. When Dr. Alexandre Almeida and his team at the University of Cambridge analyzed gut microbiome samples from more than 11,000 people across 39 countries, CAG-170 kept appearing with striking consistency: elevated in healthy individuals, diminished in those with diseases ranging from Crohn's to colorectal cancer to Parkinson's. The finding, published in Cell Host & Microbe in February 2026, suggests that some of the most important players in human health have been invisible to science for decades, not because they're rare, but because we've been looking for them the wrong way.
The implications reach far beyond a single bacterial group. If a microbe this widespread and this health-correlated has escaped notice until now, it raises a question that should unsettle anyone who thinks we understand the gut: what else have we missed?
The 3,000-Species Blind Spot
For most of microbiology's history, studying bacteria meant growing them. You'd take a sample, smear it on a nutrient plate, incubate it, and examine whatever colonies appeared. This approach built the foundation of modern medicine, identifying pathogens from E. coli to Staphylococcus aureus. But it also introduced a massive bias: we only studied what we could cultivate. The technique was like judging an ocean's biodiversity by cataloging only the fish that swim into a net.
Metagenomics changed the game. Instead of trying to grow individual species, researchers extract all the DNA from a sample, sequence it simultaneously, and then computationally separate the genetic material into distinct organisms. When Almeida's team applied this approach to their Unified Human Gastrointestinal Genome catalogue, they identified over 4,600 bacterial species living in human guts, and more than 3,000 of them had never been described before. These weren't exotic rarities found in a single population. Many appeared consistently across multiple countries and continents.

CAG-170 emerged from this hidden majority. The name itself reflects the bacteria's obscurity: "CAG" stands for co-abundance gene group, a technical classification for organisms known only through shared genetic patterns. These bacteria have no common name, no established role in textbooks, and until recently, no known function. They exist as sequences in a database, ghosts in the machine of the human gut.
What made them stand out was their statistical fingerprint. Across the 11,000-plus samples spanning 39 countries, primarily from Europe, North America, and Asia, CAG-170 levels were consistently higher in healthy individuals than in people diagnosed with any of 13 different chronic conditions. The correlation held regardless of geography, diet, or ethnicity. In the world of microbiome research, where findings often fail to replicate across populations, that kind of global consistency is unusual.
What CAG-170 Actually Does
The team's genetic analysis revealed two capabilities that help explain CAG-170's health association. First, the bacteria carry genes for producing substantial quantities of vitamin B12, one of the most metabolically important vitamins in human biology. Second, they possess enzymes capable of breaking down a diverse range of carbohydrates, sugars, and fibers.
But here's where the story takes an unexpected turn. The vitamin B12 that CAG-170 produces doesn't appear to benefit the human host directly. Instead, the evidence suggests it feeds other beneficial bacteria in the gut ecosystem. Think of CAG-170 as the quiet infrastructure worker of the microbiome: it doesn't perform the headline-grabbing functions itself, but it creates the conditions under which other organisms can thrive. As Almeida put it, "CAG-170 bacteria appear to be key players in human health, likely by helping us digest food components and keeping the whole microbiome running smoothly."
This nutrient-sharing model challenges a common misconception about gut bacteria. Popular science often frames the microbiome as a collection of individual species, each performing a specific job: this one makes serotonin, that one breaks down fiber, another one fights pathogens. The reality is more like an economy, where some organisms produce raw materials that enable others to function. CAG-170's role as a B12 supplier positions it as a foundational species, one whose absence might not cause a single identifiable disease but whose presence helps the entire system maintain equilibrium.
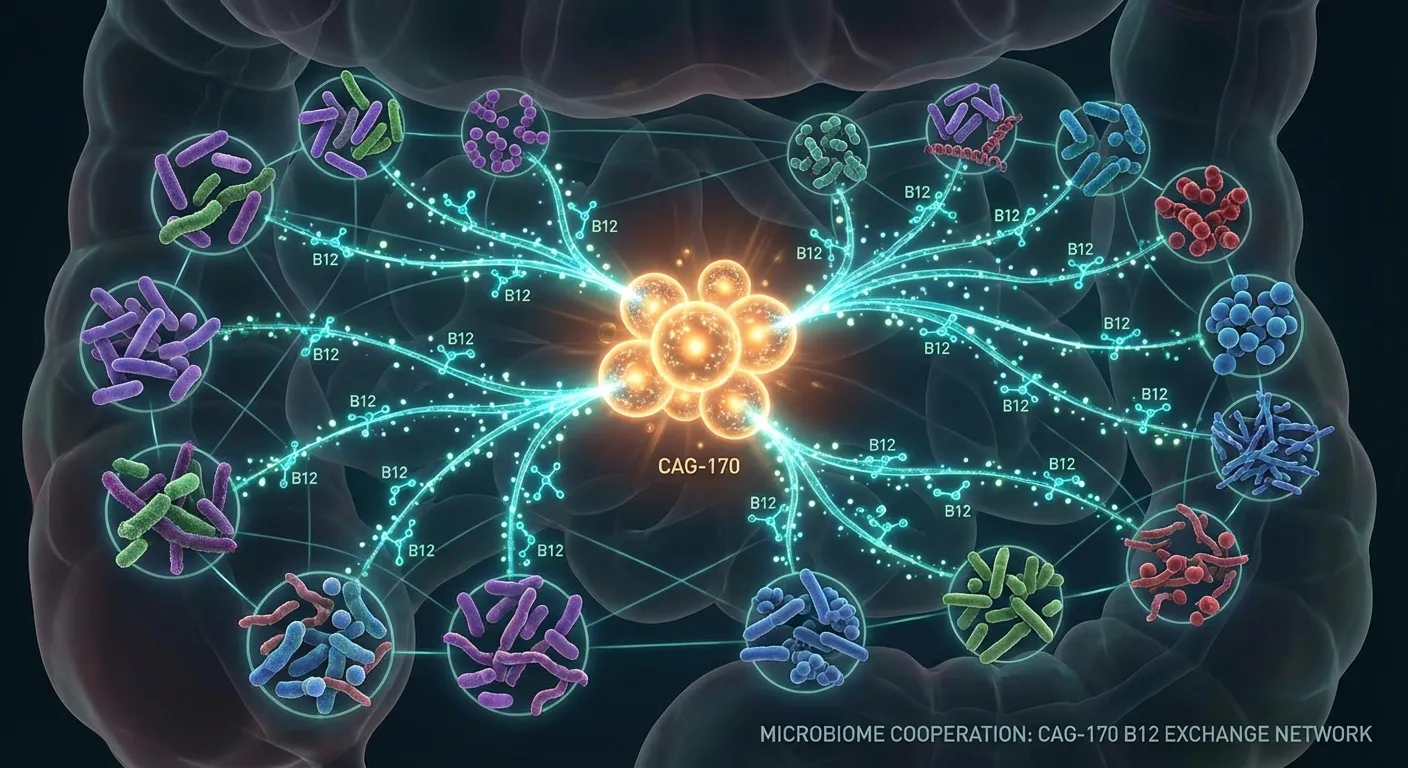
The parallel to ecology is instructive. In environmental science, ecologists have long recognized "keystone species," organisms whose influence on an ecosystem far exceeds what their abundance alone would predict. Remove the sea otter from a kelp forest, and the entire system collapses as sea urchins overgraze. Remove wolves from Yellowstone, and riverbanks erode as elk overgraze willows. CAG-170 may represent a microbial keystone species, one whose B12 production and carbohydrate processing sustain a web of downstream organisms that collectively maintain gut health. If this framework holds, it would explain a puzzle that has haunted microbiome research for years: why broad-spectrum antibiotics sometimes trigger health problems far beyond the infection they were prescribed to treat. Wiping out a keystone species, even temporarily, could cascade through the entire ecosystem.
The Probiotic Industry's Outdated Playbook
Almeida's research also delivered a pointed critique of the commercial probiotic market. "The probiotic industry hasn't really kept up with gut microbiome research," he noted. "People still use probiotic species from decades ago." Most commercially available probiotics contain Lactobacillus or Bifidobacterium strains identified in the early twentieth century. These organisms are easy to culture, easy to manufacture, and well-characterized in safety studies. What they are not, necessarily, is representative of the bacteria that actually drive gut health in 2026.
The gap between probiotic marketing and microbiome science has been widening for years, but CAG-170 makes it concrete. Here is a group of bacteria more strongly associated with health across global populations than many of the species you'd find in a grocery store supplement, yet no probiotic manufacturer can put it in a capsule. The reason is straightforward: because CAG-170 can't be cultured in a lab, there's currently no way to mass-produce it. You can't sell what you can't grow.
This creates a genuine scientific bottleneck. Before CAG-170 can be developed into a therapeutic intervention, researchers must first figure out how to cultivate it outside the human gut. That might require replicating the specific chemical environment, oxygen levels, and microbial neighbors it depends on, a challenge that has stalled development of dozens of promising uncultured organisms over the past decade. Some researchers are exploring alternative approaches, like fecal microbiota transplantation or synthetic biology, but these remain years away from clinical application.

Thirteen Diseases, One Pattern
The breadth of diseases linked to reduced CAG-170 levels is perhaps the study's most striking finding. Inflammatory bowel disease, obesity, chronic fatigue syndrome, Crohn's disease, colorectal cancer, Parkinson's disease, and multiple sclerosis all showed the same signature: less CAG-170 than healthy controls. These conditions span different organ systems, different causes, and different patient demographics. What could connect them?
The answer may lie in the ecosystem model. If CAG-170 functions as a keystone species whose B12 production and carbohydrate metabolism support broader microbial diversity, then its depletion wouldn't cause any single disease. It would create conditions of microbial instability, a weakened ecosystem more vulnerable to the specific insults that trigger different conditions in different people. This is analogous to how soil degradation doesn't cause one specific crop failure; it makes all crops more vulnerable to drought, pests, and disease.
The connection to oral bacteria and neurodegenerative disease is particularly intriguing. If microbial communities throughout the body function as interconnected ecosystems, then disruptions in one community, whether oral or intestinal, might cascade into systemic effects. The appearance of Parkinson's in CAG-170's disease list adds another data point to the growing evidence that neurological conditions have microbial dimensions science is only beginning to map.
It's worth noting a limitation, though. The study demonstrates correlation, not causation. People with chronic diseases often take medications, eat differently, and experience physiological changes that could independently reduce CAG-170 levels. The bacteria might be a casualty of disease rather than a contributor to health. Distinguishing cause from effect will require longitudinal studies tracking CAG-170 levels in healthy populations before disease onset, research that Almeida's team has indicated is already being planned.
Where This Leads
The CAG-170 discovery arrives at a moment when microbiome science is undergoing a conceptual shift. The early excitement about individual bacterial species, the "good bug, bad bug" framing, is giving way to an ecological understanding where relationships between organisms matter more than any single species. Much like the realization in ecology that organisms shape their environments in ways far more complex than simple predator-prey dynamics, microbiome research is discovering that the gut's invisible inhabitants form networks of mutual dependency that can't be reduced to a probiotic pill.
What makes CAG-170 compelling isn't just what it does. It's what it represents: an entire category of organisms, thousands strong, that have been shaping human health for millennia while remaining invisible to the methods we used to look for them. The Unified Human Gastrointestinal Genome catalogue now contains over 4,600 species, and most of them remain poorly understood. Each one might turn out to be another piece of a puzzle we didn't know we were solving.
The immediate next steps are technical: learning to culture CAG-170, confirming its causal role through animal models, and eventually testing whether restoring it improves health outcomes in depleted populations. But the deeper lesson is epistemological. For decades, microbiology defined knowledge as "what we can grow in a dish." Metagenomics has revealed that definition to be dangerously narrow. The most health-relevant bacteria on the planet might be the ones no laboratory has ever successfully kept alive outside a human body.
Sources
- University of Cambridge: "Hidden bugs in our gut appear key to good health, finds global study"
- Cell Host & Microbe: "Meta-analysis of the uncultured gut microbiome across 11,115 global metagenomes reveals a candidate signature of health"00038-7)
- Earth.com: "Mysterious gut bacteria strongly linked to human health worldwide"
- ScienceDaily: "Scientists discover a hidden gut bacterium linked to good health"