A rat encounters cat urine and, instead of fleeing in terror as every instinct should dictate, approaches it with something resembling curiosity. An ant abandons its colony at midday, climbs a plant stem to a precise height, clamps its mandibles onto a leaf vein, and waits to die. A cockroach, still alive and physically capable of running, docilely follows a wasp into a burrow where it will be eaten alive. A cricket, an insect that normally avoids water, hurls itself into a stream.
None of these animals are behaving normally. All of them have been hijacked. Their nervous systems have been infiltrated, reprogrammed, and redirected by parasites that have evolved, over millions of years, exquisitely specific mechanisms for controlling the behavior of other organisms. The science of parasitic manipulation is one of the most remarkable and unsettling corners of biology, and it is forcing researchers to reconsider fundamental questions about what behavior actually is, where it originates, and who is really in control.
The Cat Trap
The single-celled parasite Toxoplasma gondii infects an estimated one-third of the world's human population, but humans are accidental hosts. Toxoplasma's life cycle depends on cats: the parasite can only reproduce sexually in the feline intestine. Cats shed Toxoplasma oocysts in their feces, which are ingested by rodents, which are eaten by cats, completing the cycle. The problem, from the parasite's perspective, is that rodents are terrified of cats and take elaborate measures to avoid them.
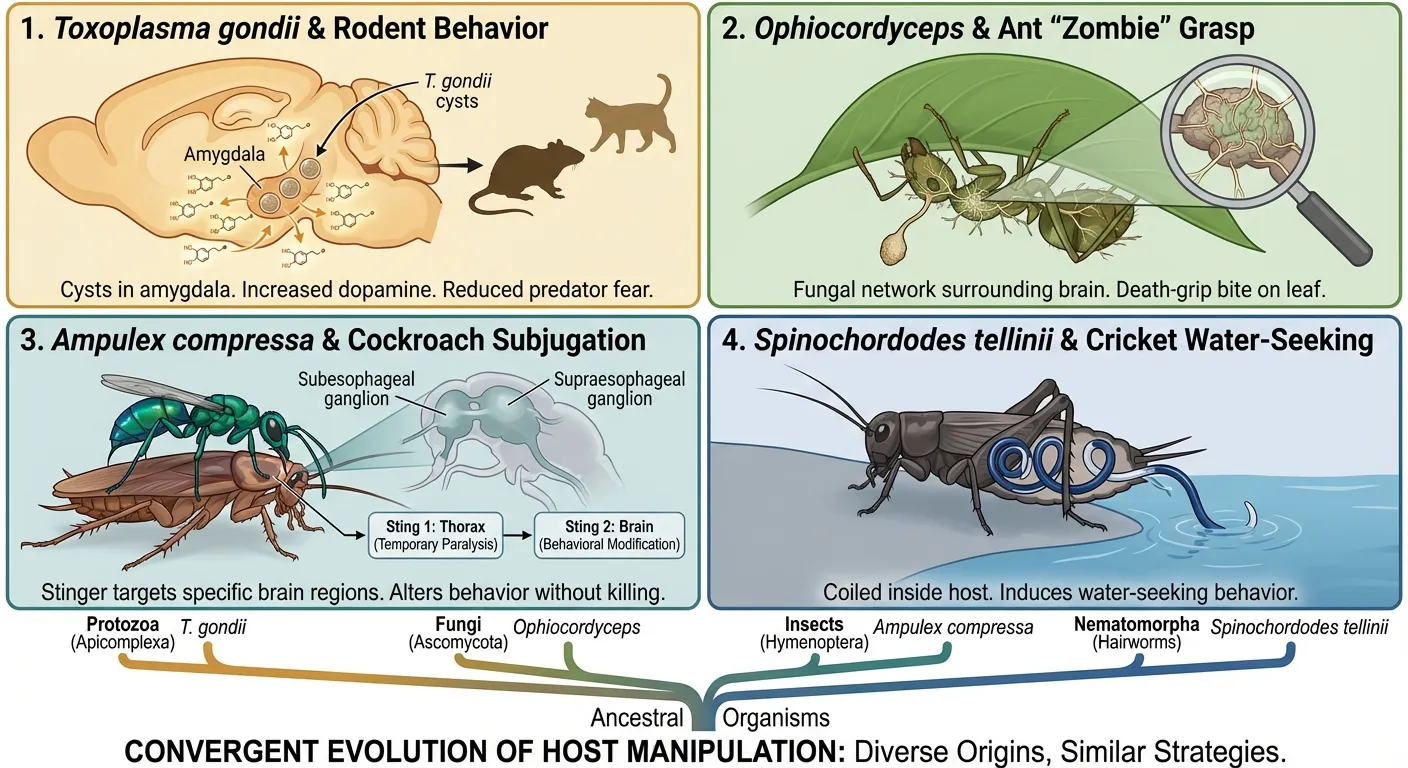
Toxoplasma's solution is elegant and disturbing. In infected rodents, the parasite forms cysts primarily in the brain, with a density in the amygdala (the brain's fear-processing center) roughly twice that of other brain regions. Infected rats don't just lose their innate aversion to cat urine. They become actively attracted to it. A 2007 study published in Proceedings of the National Academy of Sciences demonstrated that this behavioral change is remarkably specific: infection did not reduce learned fear or general anxiety. The parasite altered one precise behavioral response, the innate aversion to predator odor, while leaving everything else intact.
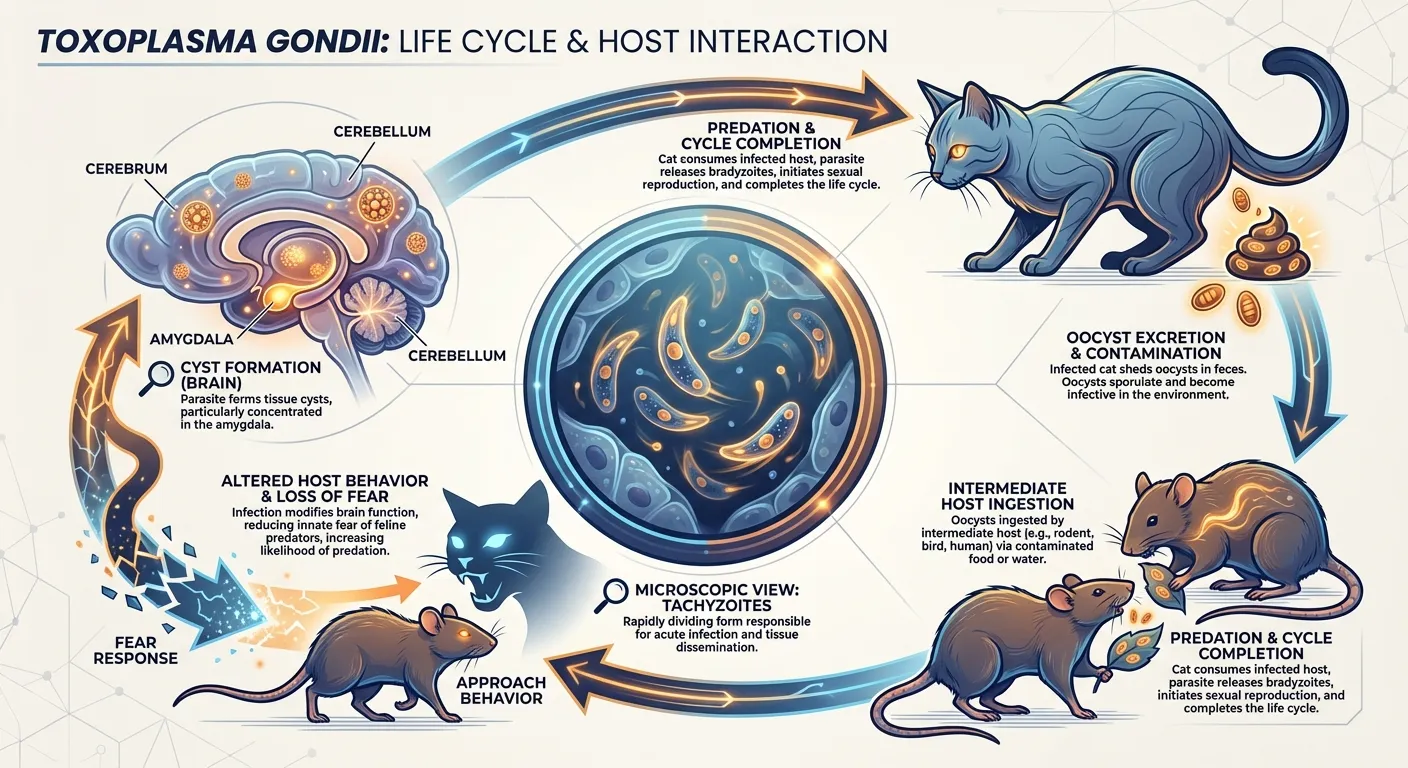
The mechanism appears to involve the parasite's own production of neurotransmitters. A 2025 study published in Nature Communications provided direct evidence that Toxoplasma produces tyrosine hydroxylase (TgTH), an enzyme that synthesizes dopamine, and that the levels of this enzyme correlate with the degree of behavioral alteration in infected rats. The parasite is, in a meaningful biochemical sense, drugging its host with dopamine, the neurotransmitter associated with reward and motivation, in the specific brain region responsible for fear of predators. The rat doesn't just stop fearing cats. It is rewired to find the scent of its predator chemically rewarding.
The implications for humans remain debated but intriguing. Studies have found correlations between Toxoplasma infection and increased risk-taking behavior, altered reaction times, and even higher rates of traffic accidents, though causation is difficult to establish in human populations. What is clear is that a single-celled organism, with no nervous system of its own, has evolved the capacity to manipulate a mammalian brain with surgical precision.
The Zombie Maker
If Toxoplasma is a subtle manipulator, Ophiocordyceps unilateralis is a blunt instrument of terrifying sophistication. This parasitic fungus infects carpenter ants in tropical forests and transforms them into what researchers have accurately termed "zombies."
The infection begins when a fungal spore lands on an ant and penetrates its exoskeleton. Over the following two to three weeks, the fungus grows inside the ant's body, surrounding and invading muscle fibers throughout the host. Fungal cells form a three-dimensional network within the ant's tissues, and researchers at Penn State University discovered something remarkable: the fungus does not invade the brain. Instead, it surrounds it, controlling the ant's behavior by manipulating its muscles and body chemistry while leaving the brain itself structurally intact. The ant's brain is, in effect, trapped inside a body it no longer controls.
The behavioral manipulation follows a precise script. The infected ant abandons its normal routine, leaves the colony at an abnormal time (healthy carpenter ants do not forage during midday), and climbs down from the canopy to a specific height, usually about 25 centimeters above the forest floor, where humidity and temperature conditions are optimal for fungal growth. There, the ant performs a "death grip," clamping its mandibles onto a leaf vein with a force that persists even after death. The fungus then erupts from the ant's head, growing a stalk topped by a fruiting body that rains spores onto the forest floor below, where they will infect the next generation of ants.
The precision of this manipulation is staggering. The ant doesn't just climb and bite any surface. It navigates to a specific microhabitat, at a specific time, and executes a specific mechanical action. As of 2023, researchers have identified at least 35 species of Ophiocordyceps capable of this kind of behavioral control, with each species typically specializing in a particular host ant species, and the actual number is likely in the hundreds.
The Neurosurgeon Wasp
The emerald jewel wasp, Ampulex compressa, takes a different approach to host control. Rather than the slow infiltration of Toxoplasma or the total-body takeover of Ophiocordyceps, the jewel wasp performs what can only be described as field neurosurgery.

When the wasp encounters a cockroach, it delivers two stings in rapid succession. The first targets the prothoracic ganglion in the cockroach's thorax, temporarily paralyzing the front legs and buying the wasp time for its second, more precise injection. The second sting is guided by sensory organs on the wasp's stinger that can feel the texture of brain tissue, allowing it to locate and penetrate specific regions of the cockroach's brain. The wasp injects venom containing GABA, beta-alanine, and taurine directly into the brain's escape circuitry.
The result is not paralysis. The cockroach can still walk, still sense its environment, still respond to stimuli at a basic level. What it loses is the will to escape. Sensory signals that would normally trigger evasive behavior still reach the cockroach's brain, but they no longer evoke a behavioral response. The cockroach becomes, in the words of researchers studying the phenomenon, "a passive zombie, bereft of free will."
The wasp then leads the cockroach into a burrow by gripping what remains of one of its antennae (the wasp has chewed the other one off to drink hemolymph for energy). Inside the burrow, the wasp lays an egg on the cockroach's abdomen. When the larva hatches, it feeds on the cockroach, which remains alive and passive throughout, eventually consuming it entirely. The wasp larva pupates inside the cockroach's hollowed-out body and emerges as an adult.
The Drowning Wire
The hairworm Spinochordodes tellinii presents perhaps the most mechanistically puzzling case of parasitic manipulation. This nematomorph parasite develops inside crickets and grasshoppers but needs to reach water to complete its life cycle and reproduce. The problem is that crickets are terrestrial insects with no reason to seek out water and good reason to avoid it (they drown easily).
Infected crickets, however, actively seek water and jump into streams, ponds, or even swimming pools. The hairworm, which by this point has grown to fill most of the cricket's body cavity, then emerges and swims away to mate, leaving the cricket to drown.
Research into the mechanism reveals that the hairworm produces proteins that mimic the cricket's own neurochemicals. A proteomic analysis found that S. tellinii secretes proteins linked to neurotransmitter release and others associated with geotaxis (movement in response to gravity), essentially reprogramming the cricket's orientation behavior to drive it toward water. The manipulation appears to involve the worm hijacking the cricket's own signaling molecules rather than introducing foreign chemicals, making the takeover particularly difficult for the host's immune system to detect or resist.
Convergent Solutions to the Same Problem
What makes parasitic manipulation so scientifically significant is not just its dramatic individual cases but the pattern that emerges across them. These parasites are separated by vast evolutionary distances. Toxoplasma is a protozoan. Ophiocordyceps is a fungus. Ampulex is an insect. Spinochordodes is a nematomorph. They share no recent common ancestor, and their manipulation strategies evolved independently. Yet they have converged on remarkably similar solutions.
All four target their hosts' nervous systems. All four alter specific behaviors while leaving general function intact (the host remains alive and mobile, which is essential for the parasite's reproductive strategy). And all four exploit the hosts' own neurochemistry, either by producing the hosts' neurotransmitters (Toxoplasma and dopamine), by physically manipulating neural and muscular systems (Ophiocordyceps and Ampulex), or by mimicking the hosts' signaling molecules (Spinochordodes).
This convergence tells evolutionary biologists something important: the nervous system is not just a target of parasitic manipulation but an inherently manipulable system. The same features that make nervous systems flexible and adaptable, their reliance on chemical signaling, their modular organization, their capacity for learning and behavioral change, also make them vulnerable to exploitation by organisms that have had millions of years to probe their weak points.
The research also has practical implications. The remarkable cognitive abilities of animals like crows demonstrate how sophisticated neural architectures can be, but parasites reveal the other side of that coin: sophisticated systems have sophisticated vulnerabilities. Understanding how parasites manipulate neurotransmitter systems could inform pharmaceutical design. If a single-celled protozoan can target dopamine signaling in a specific brain region to alter a specific behavior, researchers might learn to do the same with therapeutic precision. Several research groups are already studying the jewel wasp's venom as a potential source of novel neuroactive compounds.
The Bigger Picture
Parasitic manipulation challenges one of our most fundamental assumptions about animal behavior: that an organism's actions serve its own interests. When an infected rat approaches a cat, whose interest is being served? The rat's brain is processing sensory information and generating motor commands, but the behavioral output benefits the parasite, not the host. The hardware belongs to the rat. The software has been rewritten by Toxoplasma.
This raises uncomfortable questions that extend beyond parasitology. If a microorganism with no brain can alter the decision-making of an organism with one, what does that say about the nature of behavioral control? The neuroscience of memory and forgetting has revealed that even our own brains actively edit our mental lives in ways we don't consciously direct. Parasites simply demonstrate an external version of the same principle: that behavior is the product of biochemical processes, and those processes can be influenced by agents other than the self.
An estimated 40 percent of known animal species are parasites, and the majority of parasite-host interactions have never been studied in detail. The cases described here, Toxoplasma, Ophiocordyceps, Ampulex, Spinochordodes, are almost certainly a small fraction of the behavioral manipulation strategies that exist in nature. As researchers develop more sophisticated tools for studying neural circuits and molecular signaling in vivo, the list of known manipulators is likely to grow substantially.
What will not change is the central insight: that the line between an organism's own behavior and externally induced behavior is far blurrier than common sense suggests. Every animal navigates its world using a nervous system that can be hacked, and millions of years of evolution have produced an extraordinary diversity of hackers.
Sources
- Behavioral changes induced by Toxoplasma infection of rodents are highly specific to aversion of cat odors (PNAS, 2007)
- The role of parasite-produced dopamine in Toxoplasma gondii-altered host behaviour (Nature Communications, 2025)
- 'Zombie ant' brains left intact by fungal parasite (Penn State University)
- Parasites: evolution's neurobiologists (Journal of Experimental Biology)
- How a Wasp Turns Cockroaches into Zombies (Scientific American)

