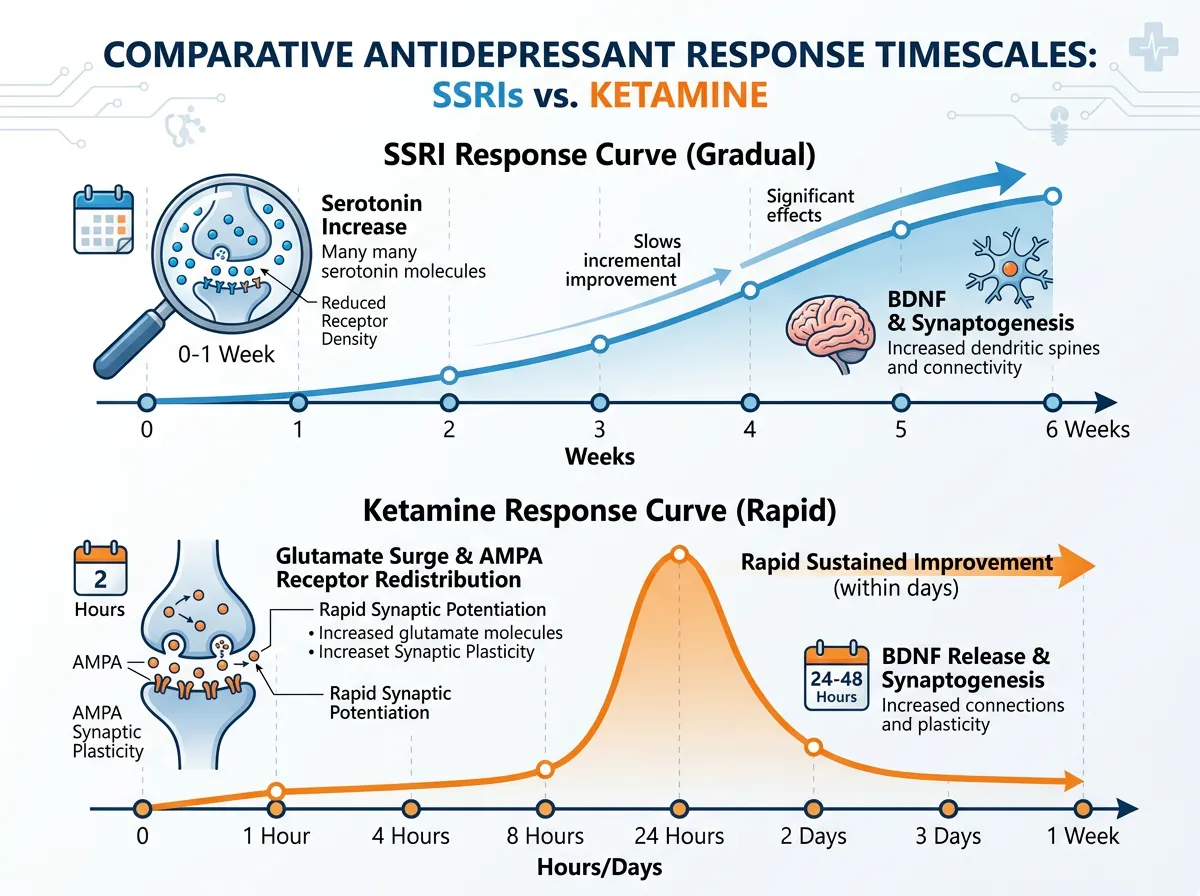
Ketamine was an anesthetic before it was a party drug and a party drug before it became one of the most promising treatments for severe depression. The trajectory is strange, but the clinical results aren't in dispute: ketamine can lift the symptoms of treatment-resistant depression within hours, sometimes minutes, in patients who have tried and failed multiple conventional antidepressants over years. What nobody has been able to adequately explain is why. Standard antidepressants take weeks to work. Ketamine works in hours. The mechanism had to be fundamentally different, but exactly how it differed remained unclear.
A study published in Molecular Psychiatry on March 5 has brought that picture into focus for the first time. Researchers in Japan used positron emission tomography, PET scanning, to directly observe what ketamine does to a specific type of receptor in the brains of people with treatment-resistant depression. The images show receptor density increasing in some regions and decreasing in others, and those changes map precisely onto which patients got better and which didn't. After decades of knowing that ketamine works without fully understanding how, science now has something closer to a wiring diagram.
What the Researchers Actually Measured
The study, led by a team across three Japanese clinical trials, focused on a protein called the AMPA receptor (technically the glutamate alpha-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid receptor, which is why everyone calls it AMPAR). This receptor sits on the surface of neurons and plays a central role in synaptic plasticity, the brain's ability to strengthen or weaken connections between nerve cells. Synaptic plasticity is the physical basis of learning, memory, and mood regulation.
The researchers used a radioactive tracer that binds specifically to AMPA receptors, then tracked it with PET scans before and after a two-week course of intravenous ketamine. The study included 34 patients with treatment-resistant depression and 49 healthy control participants. Half the patients received ketamine; the other half received a placebo.
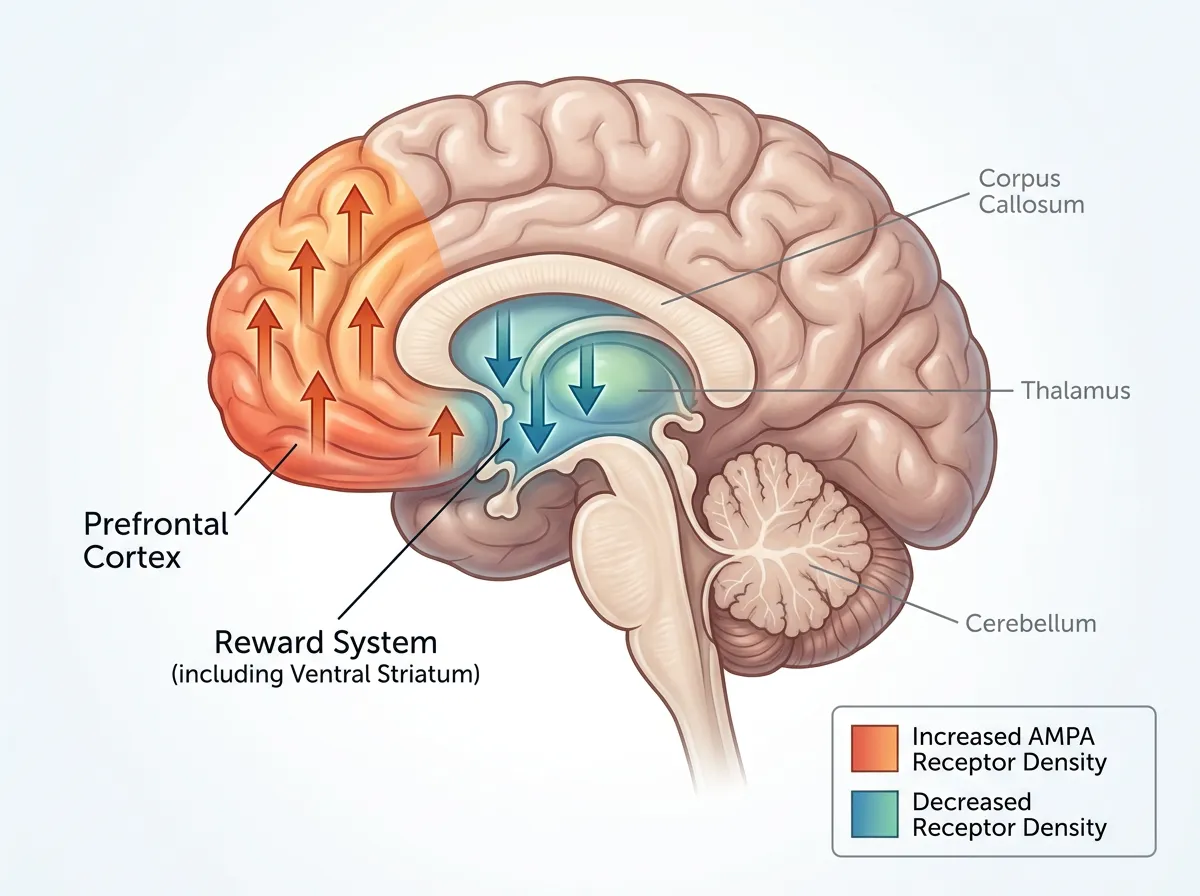
What they found was both specific and surprising. In the cortical regions of the brain, areas associated with executive function, planning, and emotional regulation, AMPA receptor density increased after ketamine treatment. In the reward-related regions deeper in the brain, areas tied to motivation, pleasure, and the ability to feel interest in things, receptor density decreased. The pattern wasn't random. It was region-specific, and it correlated tightly with symptom improvement. Patients whose receptor changes were most pronounced were the ones who reported the greatest relief from depressive symptoms.
Why AMPA Receptors, Not Serotonin, Are the Story
For the past 40 years, the dominant theory of depression has centered on serotonin. Selective serotonin reuptake inhibitors, the SSRIs that include drugs like Prozac, Zoloft, and Lexapro, work by increasing serotonin levels in the spaces between neurons. The theory was straightforward: depression results from too little serotonin, so increasing serotonin availability should fix the problem.
It was never that simple. SSRIs increase serotonin levels within hours of the first dose, but the clinical effect takes two to six weeks to appear. Something else is happening in that gap, and researchers have spent decades trying to figure out what. The serotonin hypothesis also doesn't explain why roughly 30% of depressed patients don't respond to any serotonin-based treatment, a population clinically defined as having treatment-resistant depression.
Ketamine works through an entirely different neurotransmitter system: glutamate, the brain's primary excitatory chemical messenger. Glutamate signaling is far more fundamental to brain function than serotonin. While serotonin modulates mood, glutamate drives the basic communication between neurons that underlies all cognitive and emotional processing. When you strengthen or weaken a synaptic connection (synaptic plasticity), glutamate and its receptors, including AMPA, are the primary machinery doing the work.
The Japanese PET study provides the first direct human evidence that ketamine's antidepressant effect operates through AMPA receptor redistribution, not just a generalized increase in glutamate activity. Previous research using animal models had suggested AMPA involvement, but those studies couldn't measure receptor density changes in specific brain regions of living humans. This study can, and what it shows is that ketamine doesn't just flood the brain with glutamate. It reorganizes where the brain is most responsive to glutamate signaling, shifting sensitivity toward cortical control regions and away from reward circuits.
What "Treatment-Resistant" Actually Means
To appreciate why this research matters clinically, you have to understand what the 34 patients in this study had been through before they received ketamine. Treatment-resistant depression isn't a diagnosis you receive at your first appointment. It's a classification earned through failure: typically, two or more adequate trials of different antidepressant medications at therapeutic doses for appropriate durations, none of which produced meaningful symptom relief.
For many patients, the journey to a treatment-resistant diagnosis spans years. They try an SSRI. It doesn't work, or the side effects are intolerable. They try another. Same result. They try an SNRI (serotonin-norepinephrine reuptake inhibitor), a tricyclic antidepressant, possibly a monoamine oxidase inhibitor. They may try psychotherapy, exercise programs, dietary changes, and supplements. By the time a patient is classified as treatment-resistant, they have often spent three to five years in a cycle of hope and disappointment that compounds the depression itself.
These are the patients for whom ketamine represents something genuinely new. Not a slightly different formulation of the same serotonin strategy, but a fundamentally different approach that targets different receptor systems and works on a timescale measured in hours rather than weeks. The PET imaging study adds a critical dimension: it suggests that AMPA receptor patterns could serve as a biomarker, a measurable indicator that helps doctors predict who will respond to ketamine before starting treatment, rather than waiting to see if the drug works through trial and error.
The Caveats That Make This Study Honest
The authors of the Molecular Psychiatry paper are careful about what they claim, and that caution is itself informative. The study included 34 patients with treatment-resistant depression. That's enough to identify a pattern, but it's not enough to establish the kind of robust, generalizable conclusions that change clinical practice. Larger, more diverse studies are needed to confirm that the AMPA receptor changes observed in Japanese patients also appear in populations with different genetic backgrounds, medication histories, and subtypes of depression.
There's also the question of duration. Ketamine's antidepressant effects, while fast, tend to be temporary. Most patients experience relief for days to weeks after a single infusion, but the depression typically returns. Repeated infusions can extend the benefit, but the optimal dosing schedule remains an open question. The PET study measured receptor changes after a two-week treatment course. It doesn't tell us whether those changes persist, reverse, or evolve over months.
The placebo comparison adds nuance. Patients who received placebo infusions did not show the same receptor density changes, which strengthens the case that the AMPA redistribution is genuinely drug-driven and not a product of placebo response or the experience of receiving medical attention. But placebo-controlled studies of ketamine face a methodological challenge: ketamine produces noticeable dissociative effects (altered perception, floating sensations, mild hallucinations) that make true blinding difficult. Patients often know whether they received the drug, which can influence their self-reported symptom improvements.
From Brain Scan to Bedside: The Biomarker Question
The most practical implication of this research is the possibility that PET imaging of AMPA receptors could become a clinical tool, a way to identify which patients are most likely to respond to ketamine before they receive a single dose.
This matters because ketamine treatment isn't trivial. It requires supervised intravenous infusion in a clinical setting, carries risks of dissociation and elevated blood pressure, and costs several hundred dollars per session without insurance coverage. Most insurance plans cover ketamine for anesthesia but not for depression, making treatment a significant financial burden for patients already exhausted by years of unsuccessful medication trials.
If a baseline PET scan could predict, based on a patient's existing AMPA receptor distribution, whether ketamine is likely to help, it would transform the treatment from a "let's try this and see" proposition into a targeted intervention. Patients with receptor patterns that don't match the responder profile could be directed toward other emerging treatments, including psilocybin-assisted therapy, transcranial magnetic stimulation, or newer glutamate-modulating drugs currently in clinical trials.
Yale's ketamine research program, which pioneered much of the early clinical work on ketamine for depression, has called for exactly this kind of biomarker-driven approach. The Japanese PET study doesn't deliver it yet, the sample is too small and the imaging technique too expensive for routine clinical use, but it establishes the biological foundation that future, larger studies can build on.
Where This Leads
The PET imaging study joins a growing body of work that is slowly moving the field of psychiatry from symptom-based treatment to mechanism-based treatment. For most of the discipline's history, depression treatment has been essentially trial and error: prescribe a drug, wait weeks, assess, try something else if it fails. The biological reasons why one drug works for one patient and not another have been largely invisible.
AMPA receptor imaging won't replace SSRIs or make ketamine the default treatment for depression. But it introduces a level of biological specificity that the study of habits and automatic behavior has only recently achieved in adjacent fields of neuroscience. If receptor distribution patterns can predict treatment response, psychiatry gains something it has historically lacked: a way to match interventions to biology rather than guessing.
The 34 patients in the Japanese study represent a small sample but a large idea. For the first time, researchers can watch an antidepressant reshape the receptor architecture of a living human brain and correlate those changes with clinical improvement. The image is still blurry, limited by sample size and methodological constraints. But the fact that the image exists at all, that we can see where ketamine works and begin to understand why, represents a genuine shift in how neuroscience approaches one of medicine's most stubborn disorders.
Sources
- Brain scans reveal how ketamine quickly lifts severe depression - ScienceDaily
- New PET imaging study reveals how ketamine relieves treatment-resistant depression - EurekAlert
- Brain Imaging Reveals How Ketamine Treats Depression - Technology Networks
- Ketamine: How Yale Redefined Depression Treatment - Yale School of Medicine