One of the strangest facts about the universe is that matter doesn't behave the way common sense suggests. At small scales, particles like electrons act like waves, spreading out and interfering with themselves in ways that solid objects shouldn't be able to do. This wave-particle duality has been experimentally confirmed for nearly a century, but it raises an uncomfortable question: where does the quantum weirdness end and normal reality begin?
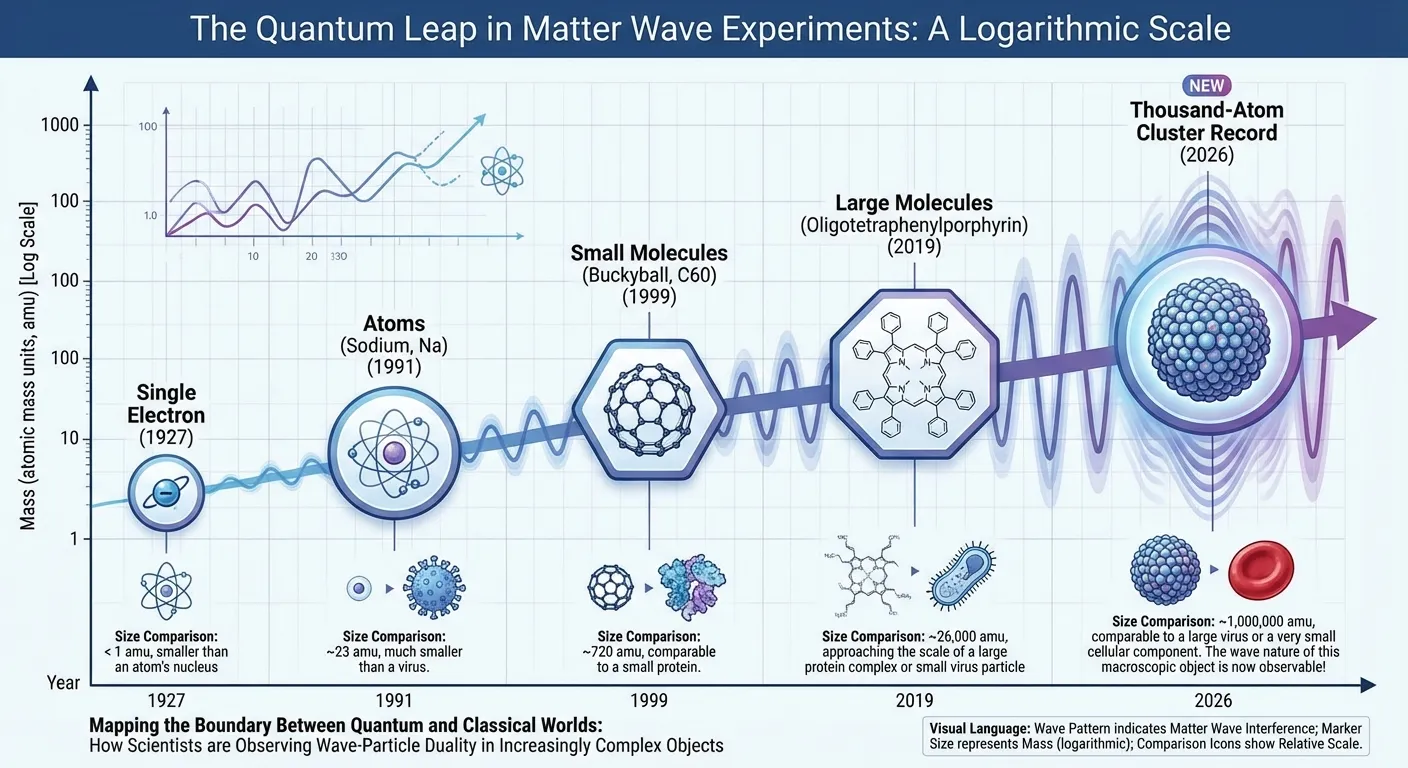
A new experiment has pushed that boundary further than ever before. A team led by Markus Arndt at the University of Vienna has coaxed a cluster containing thousands of atoms into behaving like a wave, not a collection of particles. The result didn't just break a record. It put real experimental pressure on theories that try to explain why the quantum world and the everyday world seem to operate by different rules.
Understanding what this means requires understanding what matter waves actually are, why size matters, and what the experiment reveals about the nature of reality at its most fundamental level.
What Are Matter Waves?
In 1924, a French physicist named Louis de Broglie proposed something radical. If light, which had long been understood as a wave, could also behave like particles (photons), then perhaps the reverse was also true. Perhaps particles like electrons could also behave like waves. He derived a simple equation relating a particle's wavelength to its momentum: the smaller the momentum, the longer the wavelength.
De Broglie's hypothesis was confirmed just a few years later when physicists demonstrated that electrons passing through crystals produced interference patterns, exactly as waves would but particles shouldn't. The electrons were somehow spreading out, interfering with themselves, and producing characteristic wave-like patterns. This wasn't a peculiarity of electrons; subsequent experiments showed that protons, neutrons, atoms, and even molecules exhibited the same behavior.
The de Broglie wavelength depends inversely on mass and velocity. Heavier objects have shorter wavelengths; faster objects have shorter wavelengths. For everyday objects, the wavelength is so incredibly tiny that wave behavior is completely undetectable. A baseball traveling at 100 mph has a de Broglie wavelength smaller than the diameter of an atomic nucleus. For all practical purposes, baseballs are particles, not waves.
But as objects get smaller and colder (slower), their wavelengths get longer. Cool an atom to near absolute zero, and its de Broglie wavelength can stretch to micrometers, long enough to observe directly. This is the regime where quantum mechanics becomes visible, where matter stops behaving like the solid stuff we experience and starts revealing its wave nature.
The Challenge of Demonstrating Wave Behavior
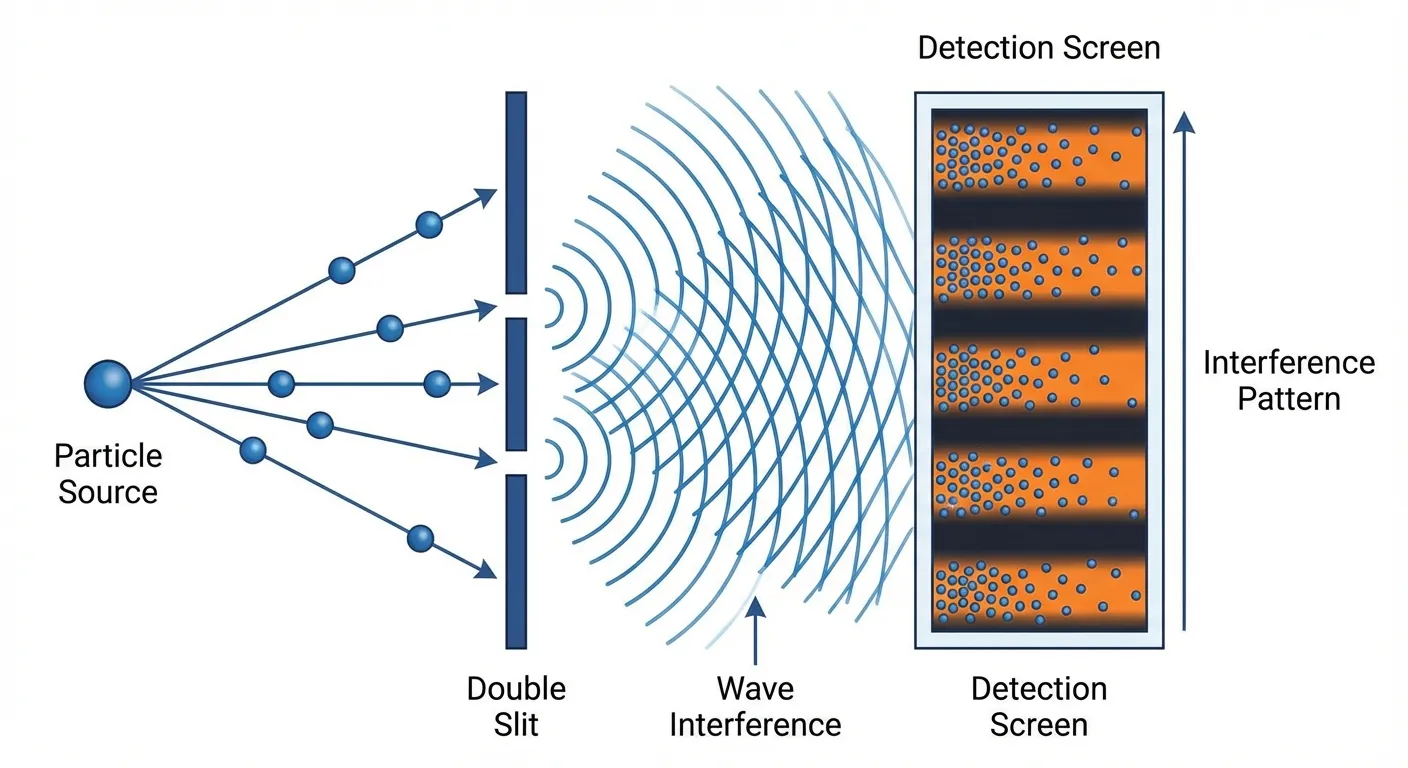
Demonstrating that something acts like a wave requires showing interference. When two waves meet, they can add together (constructive interference) or cancel out (destructive interference), creating characteristic patterns that particles simply can't produce. The classic double-slit experiment sends particles through two openings; if they're waves, they'll interfere with themselves and create a striped pattern on a detector.
The difficulty is that wave behavior is fragile. Interactions with the environment, through collisions with gas molecules, absorption or emission of light, or any other physical process, cause the wave function to "collapse" into particle behavior. The larger an object, the more ways it can interact with its environment, and the faster this decoherence occurs. Maintaining wave-like behavior requires extraordinary isolation.
Temperature is critical. Atoms at room temperature are vibrating rapidly and constantly emitting and absorbing thermal radiation. This continuous interaction with the environment destroys quantum coherence almost instantly. To observe wave behavior, researchers must cool their atoms to temperatures measured in nanokelvins, billionths of a degree above absolute zero. At these temperatures, atoms move slowly enough that their de Broglie wavelengths become measurable and they interact with their environment slowly enough that coherence can persist.
Previous experiments had demonstrated wave behavior in single atoms, small molecules, and clusters of a few hundred atoms. Each increase in size represented a technical triumph, requiring better cooling, better isolation, and more sensitive detection. The new experiment pushed this boundary dramatically further.
The Breakthrough Experiment
The experiment used sophisticated laser cooling and trapping techniques to prepare a cluster of atoms in an extremely cold, isolated state. The atoms were caught in a carefully configured electromagnetic trap that held them in place while lasers cooled them to near absolute zero. Once prepared, the trap was turned off, allowing the atoms to fall freely.

As the cluster fell, it passed through a series of laser pulses that acted like the slits in a double-slit experiment. The pulses split the atomic wave function into multiple paths, allowed the paths to evolve separately, and then recombined them. If the cluster maintained quantum coherence through this process, the recombined wave functions would interfere, producing patterns that could be detected.
The detection confirmed wave-like interference. The cluster of thousands of atoms produced the characteristic striped pattern of constructive and destructive interference, the unmistakable signature that the atoms were not following individual particle trajectories. They were spreading out, taking multiple paths simultaneously, and interfering with themselves.
The technical achievement is remarkable. The experiment required maintaining quantum coherence in a system orders of magnitude larger than previous demonstrations. Every source of decoherence had to be identified and suppressed. The vacuum had to be extraordinarily good, eliminating stray gas molecules that could scatter the atoms. The laser pulses had to be precisely timed and calibrated. The detection had to be sensitive enough to resolve the interference pattern.
Why Size Matters
The philosophical interest of this result concerns where quantum mechanics ends and classical physics begins. Our everyday experience suggests that big things behave classically: baseballs follow trajectories, cars don't tunnel through walls, cats are either alive or dead, not both. But quantum mechanics contains no built-in limit. Nothing in the equations says wave behavior should disappear above a certain size.
This creates what physicists call the "measurement problem" or the "macro-objectification problem." If quantum mechanics is universal, why don't we see quantum effects in everyday life? Various proposals have been offered: perhaps consciousness collapses wave functions, perhaps gravity causes decoherence, perhaps quantum mechanics simply doesn't apply above certain scales. Each proposal is controversial, and none has been definitively confirmed or refuted.
Experiments pushing the quantum-classical boundary help constrain these proposals. If wave behavior can be demonstrated in larger and larger systems, proposals that predict quantum mechanics breaks down at small scales become less plausible. The new experiment pushes this constraint further, showing that systems far larger than previously demonstrated can still exhibit quantum effects under the right conditions.
This doesn't mean we'll see macroscopic quantum effects in everyday life. But the result shifts the conversation in an important way. Instead of asking "at what size does quantum mechanics break down?" physicists are increasingly asking "how good does the isolation need to be?" The answer appears to be: very good, but not impossibly so. Advances in cryogenics, vacuum technology, and vibration control continue to push the feasible limit upward. Each jump in scale reveals that the quantum-classical divide is not a wall but a gradient, one that technology is learning to navigate.
The Fundamental Shift
The Vienna experiment directly constrains several competing theories about the quantum-classical boundary. Continuous Spontaneous Localization (CSL) models, which propose that wave functions collapse spontaneously at rates proportional to particle number, predicted that clusters this large should have lost coherence. The successful interference observation forces CSL advocates to push their collapse rate parameter at least an order of magnitude lower than previous estimates, narrowing the theory's viable parameter space. Gravitational decoherence models proposed by Roger Penrose, which tie collapse to gravitational self-energy differences between superposed states, remain consistent with the result but will face their own test once clusters reach approximately 10 million atomic mass units.
For quantum computing and quantum sensing, the result delivers a specific engineering insight: decoherence in these thousand-atom clusters was dominated by thermal photon emission, not by collisions or electromagnetic noise. This tells device engineers that scaling up quantum systems requires solving the thermal radiation problem first. Quantum sensors built from larger atom clusters, now demonstrated to maintain coherence, could achieve gravitational wave sensitivity improvements of roughly a factor of 100 over current atom interferometers, enough to detect signals from merging intermediate-mass black holes that existing detectors miss.
The next experimental target is a cluster in the 10,000-to-100,000 atom range, which Arndt's group estimates is achievable within five years using improved cryogenic beam sources. At that scale, the experiment would definitively rule out or confirm an entire class of objective collapse theories, much as newly theorized states of water inside ice giants require quantum-level understanding to explain. Reaching that threshold would transform the quantum-classical boundary question from philosophical debate into settled experimental physics.

