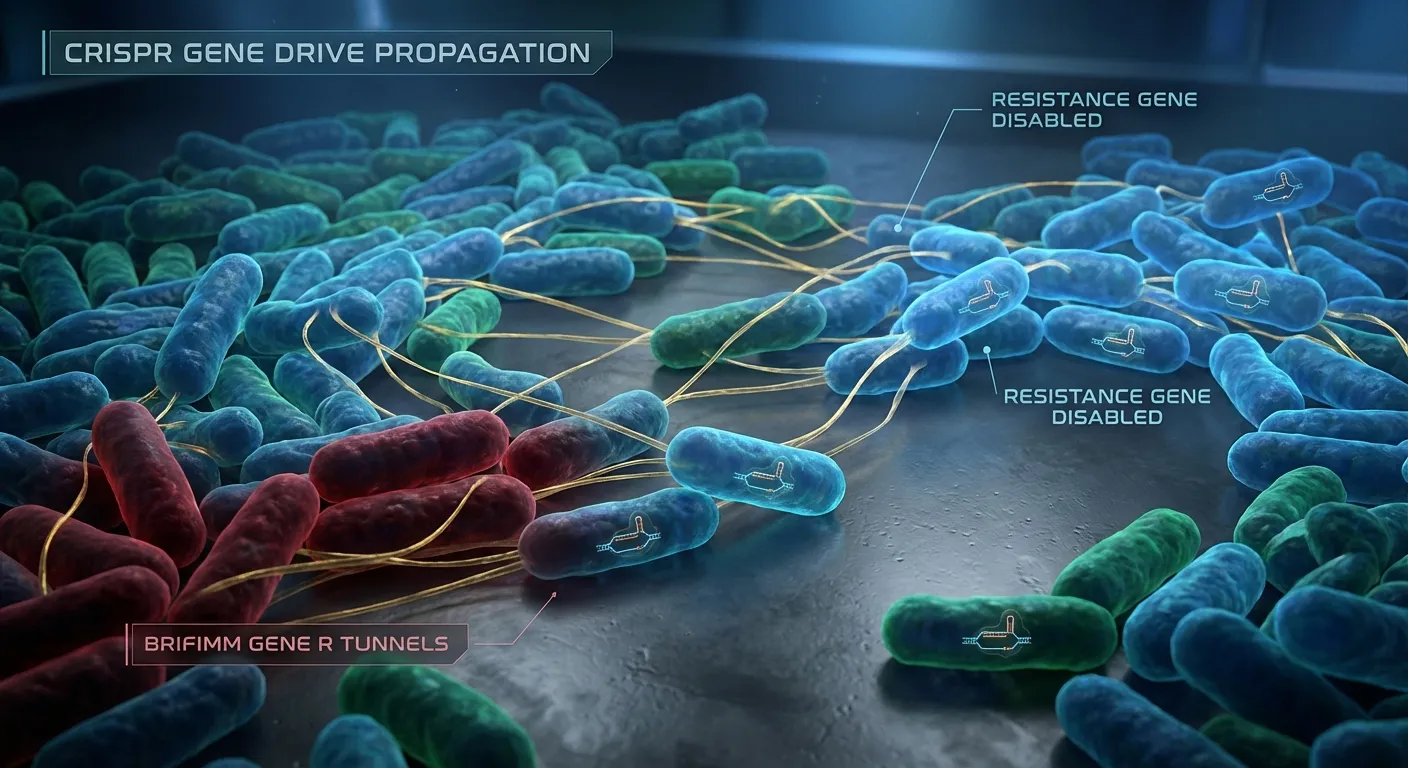
In the ongoing war between antibiotics and bacteria, bacteria have been winning. Not because they are smarter or more sophisticated, but because evolution moves faster at their scale. A single bacterium can divide every twenty minutes. Each division is an opportunity for mutation, and some of those mutations confer resistance to the drugs designed to kill them. Worse still, bacteria do not keep these advantages to themselves. They share resistance genes horizontally, passing circular DNA fragments called plasmids to neighboring cells through a process resembling mating. A resistance gene that evolves in one bacterium can spread through an entire population in hours.
The result, after decades of antibiotic overuse in medicine and agriculture, is a crisis. The World Health Organization projects that drug-resistant infections will cause more than 10 million deaths annually by 2050, surpassing cancer as a global cause of mortality. New antibiotics are not emerging fast enough to replace the ones that are failing, and the economics of drug development make it unlikely that pharmaceutical companies will close the gap on their own.
Against this backdrop, a team at UC San Diego has published something genuinely novel: a CRISPR-based system that does not kill bacteria but instead strips them of their resistance genes, turning superbugs back into ordinary, treatable infections. The technology borrows a concept from insect genetics, the gene drive, and applies it to bacterial populations for the first time with practical efficiency. If it works outside the laboratory, it could represent a fundamentally different approach to a problem that conventional medicine has been losing.
The Gene Drive Concept
To understand what the UC San Diego team built, you first need to understand gene drives, one of the most powerful and controversial tools in modern genetics.
In sexually reproducing organisms, any given gene has a roughly 50% chance of being passed to the next generation. That is standard Mendelian inheritance. A gene drive breaks this rule. It is a genetic element engineered to copy itself into both copies of a chromosome during reproduction, ensuring that it appears in nearly 100% of offspring rather than the expected 50%. Over generations, the drive spreads through a population exponentially, even if it confers no survival advantage.
Researchers have been developing gene drives in mosquitoes for years, targeting the species that transmit malaria. The idea is to release engineered mosquitoes carrying a drive that either renders the population infertile or makes them unable to carry the malaria parasite. The technology has shown promise in laboratory settings, though field deployment remains a subject of intense ethical debate.
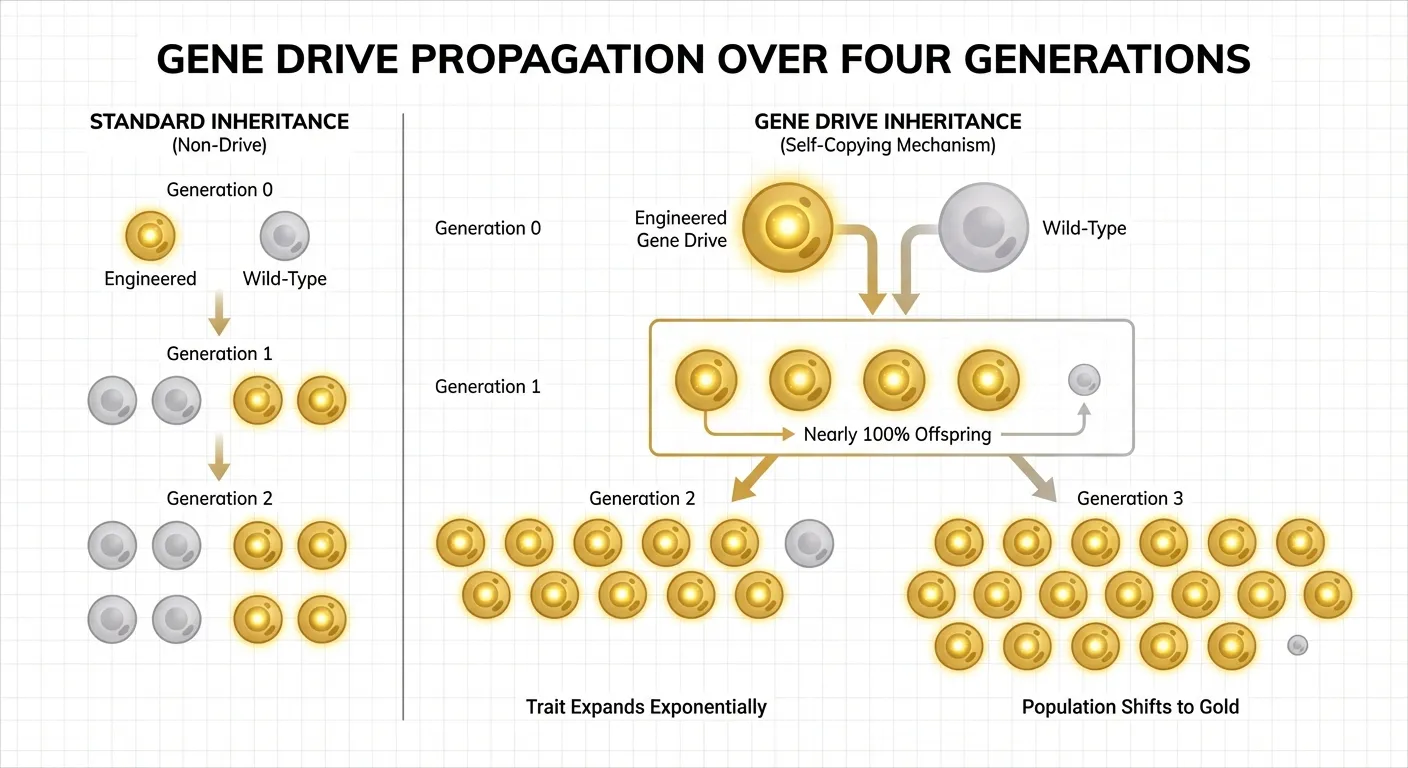
The core challenge with bacteria is that they do not reproduce sexually in the traditional sense. They divide by binary fission, cloning themselves. So a Mendelian gene drive, which depends on sexual reproduction, will not work. But bacteria have their own form of horizontal gene transfer: conjugation. During conjugation, one bacterium extends a thin tube (a pilus) to a neighboring cell and transfers a copy of a plasmid through it. This is the same mechanism that spreads antibiotic resistance so effectively. The UC San Diego team realized they could co-opt this channel to spread a CRISPR cassette that destroys resistance genes instead.
How pPro-MobV Works
The system, called pPro-MobV (Pro-Active Genetics with Mobilization V), was developed in the laboratories of professors Ethan Bier and Justin Meyer at UC San Diego's School of Biological Sciences. It builds on an earlier proof-of-concept called Pro-AG, which Bier's lab introduced in 2019 in collaboration with Victor Nizet of UC San Diego's School of Medicine.
The original Pro-AG system demonstrated that a CRISPR cassette could target and disable antibiotic resistance genes on bacterial plasmids. The cassette would locate the resistance gene, cut it using CRISPR-Cas9 molecular scissors, and insert itself into the gap, simultaneously destroying the resistance gene and copying the CRISPR system into the plasmid. The problem was delivery. The first-generation system required researchers to introduce the cassette directly into target bacteria, a process with no practical mechanism for reaching bacteria in real-world environments.
pPro-MobV solves the delivery problem by exploiting conjugation. The engineered CRISPR cassette is mounted on a plasmid that can transfer itself to neighboring cells through the same conjugal tunnels that bacteria use to share resistance genes. Once the cassette arrives in a new cell, it targets any resistance genes present, cuts them, and inserts itself. That cell then becomes a carrier, capable of passing the CRISPR cassette to its own neighbors. The system is, in effect, a contagious cure.
"With pPro-MobV we have brought gene-drive thinking from insects to bacteria as a population engineering tool," Bier explained in the study announcement.
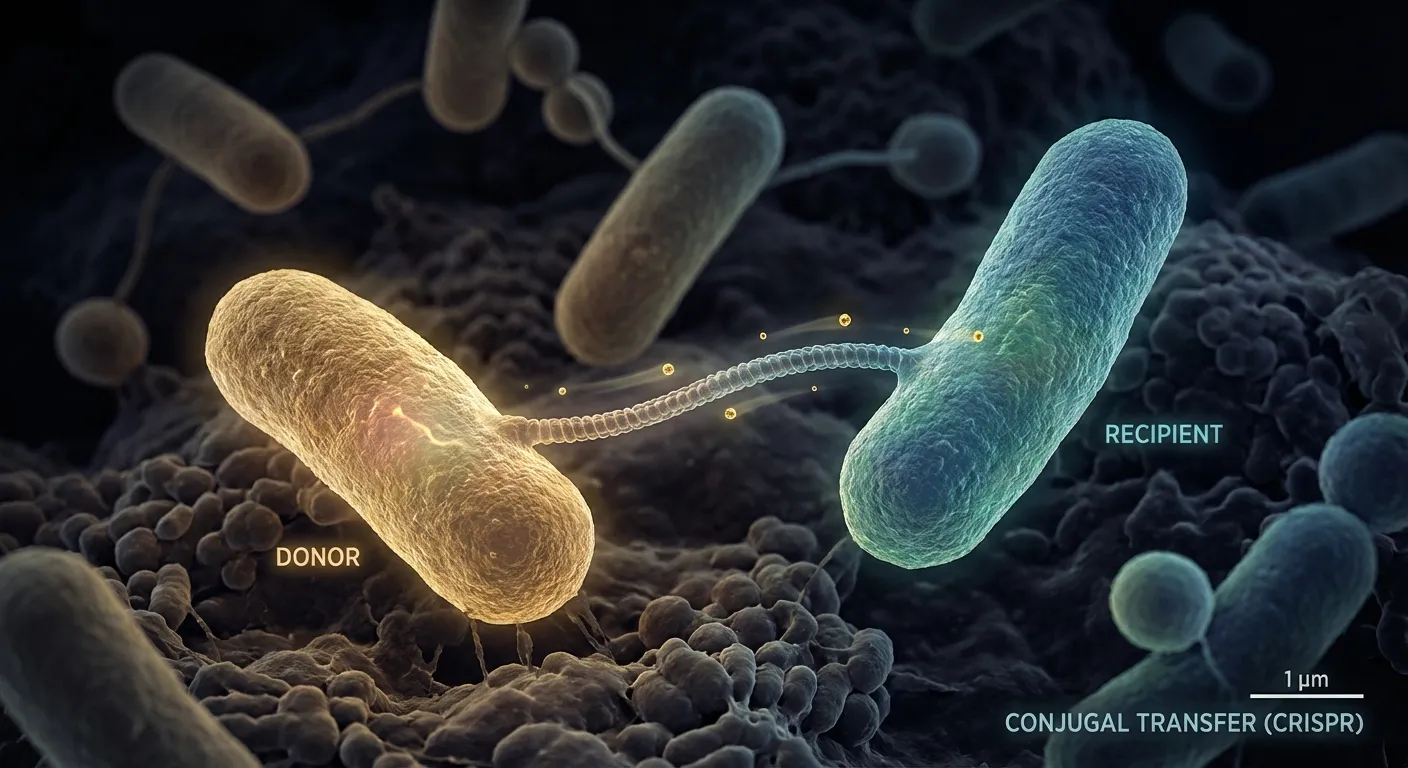
The Biofilm Breakthrough
Perhaps the most significant aspect of the new research, published in Nature npj Antimicrobials and Resistance in February 2026, is that the team demonstrated the system working within bacterial biofilms.
Biofilms are not just collections of bacteria. They are structured communities in which microorganisms embed themselves in a self-produced matrix of sugars, proteins, and DNA, forming a slimy layer on surfaces. Biofilms form on medical implants, hospital surfaces, water pipes, and wound tissue. They are notoriously difficult to treat because the matrix acts as a physical barrier, preventing antibiotics from reaching the bacteria inside. Infections involving biofilms are estimated to account for up to 80% of chronic bacterial infections in clinical settings.
Conventional antibiotics struggle with biofilms precisely because they need to penetrate the matrix to reach their targets. pPro-MobV sidesteps this problem entirely. Because it spreads through conjugation, it moves between cells that are already in physical contact within the biofilm. The matrix that blocks drugs from outside does not block genetic transfer between resident bacteria. The system works with the biofilm's internal architecture rather than against it.
This distinction is critical. Many promising antimicrobial technologies perform well in liquid cultures, where bacteria float freely and drug concentrations are uniform, but fail when confronted with the complex, structured environments where real infections occur. Demonstrating efficacy within biofilms suggests that pPro-MobV could function in the conditions that matter most clinically.
A Two-Pronged Attack: Adding Phages
The researchers also discovered that the CRISPR cassette components could be carried and delivered by bacteriophages, viruses that naturally infect and replicate within bacteria. This opens a second delivery route alongside conjugation.
Phages have their own long history in medicine. Before antibiotics became dominant in the mid-20th century, phage therapy was a standard treatment for bacterial infections in parts of the Soviet Union and Eastern Europe. The approach fell out of favor in the West after penicillin's discovery, but it has experienced a renaissance in recent years as antibiotic resistance has worsened. Several hospitals in the United States and Europe now offer phage therapy as a last resort for patients with untreatable infections.
Combining phage delivery with conjugal spreading creates a system that can attack resistance from two directions simultaneously. Phages inject the CRISPR cassette from outside the cell, while conjugation transfers it between cells that are already in contact. This dual approach makes it harder for bacteria to evolve resistance to the treatment itself, since they would need to simultaneously block both viral infection and conjugal transfer, two fundamentally different biological processes.
Justin Meyer, whose lab specializes in evolutionary dynamics, emphasized the broader significance: "This technology is one of the few ways that I'm aware of that can actively reverse the spread of antibiotic-resistant genes, rather than just slowing or coping with their spread."
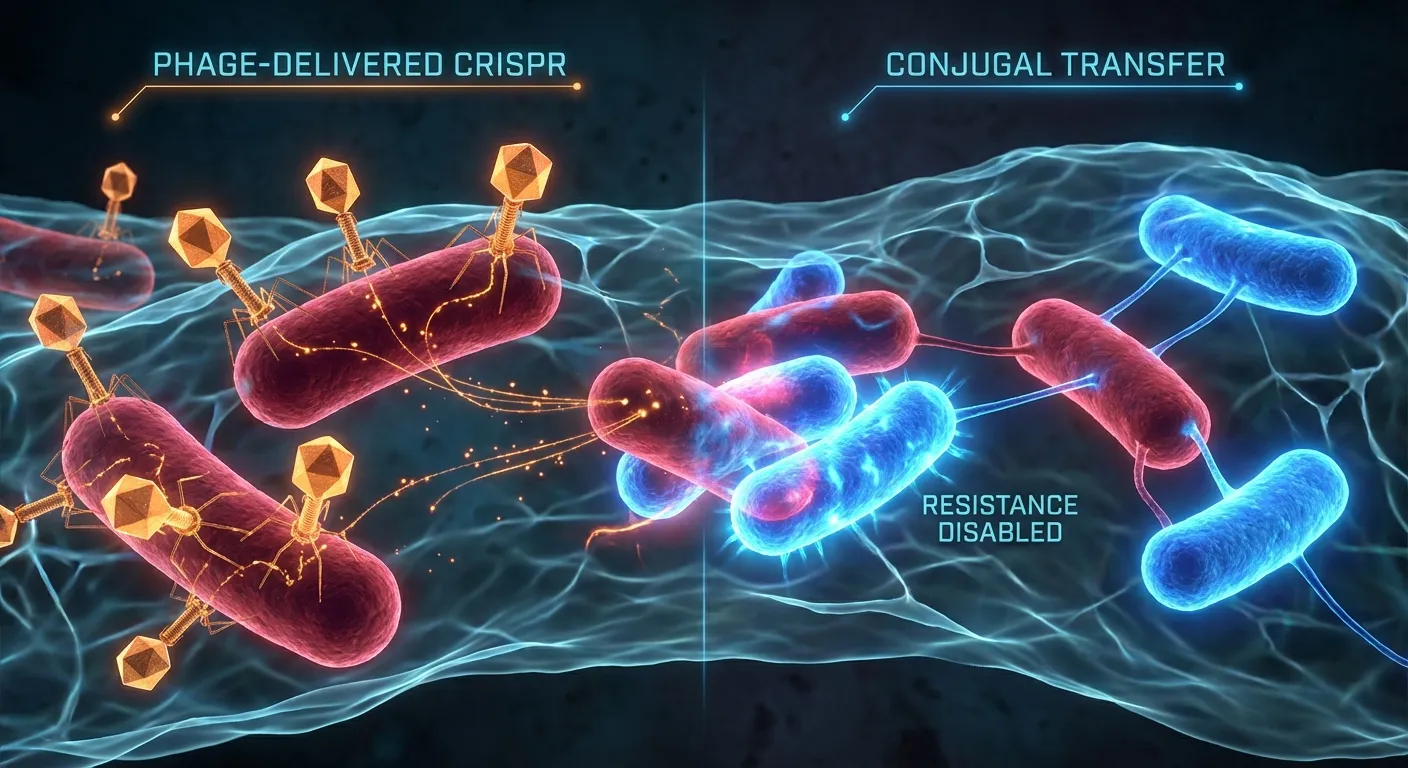
Beyond the Hospital: Environmental Applications
The implications of pPro-MobV extend well beyond clinical medicine. Researchers estimate that roughly half of antibiotic resistance originates not in hospitals but in environmental settings: aquaculture facilities, livestock operations, sewage treatment plants, and agricultural runoff. These are environments where antibiotics are used in large quantities and resistant bacteria proliferate with minimal oversight.
A system that can spread through bacterial populations and strip out resistance genes could, in principle, be deployed in these settings to reduce the reservoir of resistance before it reaches human patients. Imagine treating a fish farm's water supply or a sewage processing facility with an engineered conjugative plasmid that disables resistance genes as it spreads. The bacteria would remain alive and functional in their ecological roles, but they would lose their ability to shrug off the antibiotics that human medicine depends on.
This environmental dimension represents a conceptual shift from treating resistance as a clinical problem to treating it as an ecological one. Antibiotics work within bodies, but resistance evolves in ecosystems. A tool that operates at the population level, spreading through bacterial communities the way resistance itself spreads, matches the scale of the problem in a way that individual-patient treatments cannot.
The approach also raises questions that the research team acknowledges openly. Releasing self-spreading genetic elements into environmental bacterial populations is not without risk. The system includes a safety mechanism, a homology-based deletion feature that allows the CRISPR cassette to be removed from the population if needed. But the history of biological introductions, from cane toads in Australia to kudzu in the American South, counsels caution about self-propagating interventions in complex ecosystems. The gap between laboratory demonstration and environmental deployment will require years of additional research, regulatory development, and careful risk assessment.
Where This Leads
The pPro-MobV system represents something genuinely new in the fight against antibiotic resistance: a tool that uses bacteria's own mechanisms of gene sharing against them. Instead of trying to kill resistant bacteria with ever-stronger drugs, it infiltrates their populations and disables the very genes that make them dangerous. It is, in a sense, an informational weapon rather than a chemical one, targeting the genetic instructions for resistance rather than the bacteria themselves.
Whether this approach will succeed in the messy complexity of real-world infections remains to be seen. The jump from laboratory biofilms to hospital wards, agricultural facilities, and environmental water systems is enormous. Bacterial populations in the wild are genetically diverse, ecologically complex, and under evolutionary pressures that laboratory conditions cannot fully replicate. The bacteria will evolve countermeasures. The question is whether pPro-MobV can outpace those countermeasures or be iteratively updated to stay ahead.
What the UC San Diego work establishes, at minimum, is that the gene drive concept transfers from insects to bacteria with practical efficiency. That alone is a significant intellectual achievement, connecting two previously separate fields of genetics research. And the timing matters. The growing understanding of how bacteria organize in complex communities is revealing new vulnerabilities that population-level tools like pPro-MobV might exploit. Antibiotic resistance is not a problem that will be solved by a single breakthrough, but the toolkit for fighting it just gained a new and genuinely original instrument.
Sources
- Next Generation Genetics Technology Developed to Counter the Rise of Antibiotic Resistance - UC San Diego Today
- New CRISPR tool spreads through bacteria to disable antibiotic resistance genes - Phys.org
- Breakthrough CRISPR system could reverse antibiotic resistance crisis - ScienceDaily
- CRISPR vs superbugs: new genetic technology dismantles antibiotic resistance in bacteria - BioTechniques