For most of its history, cancer research operated under a straightforward assumption: tumors arise from within. Genetic mutations accumulate over time, cells lose their internal brakes, and uncontrolled growth follows. External factors like radiation or cigarette smoke were understood to cause those mutations, but the idea that a living organism, a bacterium already resident in your mouth, could travel to distant tissue and actively drive cancer forward? That would have sounded far-fetched a decade ago.
It doesn't anymore. A study published in January 2026 in Cell Communication and Signaling by researchers at the Johns Hopkins Kimmel Cancer Center has traced a direct line from Fusobacterium nucleatum, a rod-shaped bacterium best known for causing gum disease, to the initiation, growth, and spread of breast cancer. The findings don't just add another entry to the growing list of microbiome-cancer connections. They reveal a specific, step-by-step mechanism by which a common oral pathogen hijacks cellular repair systems, transforms how cells behave, and makes certain people, particularly those carrying BRCA1 mutations, far more vulnerable than anyone previously understood.
A Bacterium with a History
Fusobacterium nucleatum is not a newcomer to cancer research, though its connection to breast cancer is. The bacterium has been a subject of intense study since the early 2010s, when two independent research groups simultaneously identified it as unusually abundant in colorectal tumor tissue. The finding was initially met with skepticism. Correlation, after all, is not causation. Tumors create inflammatory, nutrient-rich environments that might simply attract opportunistic bacteria. Perhaps F. nucleatum was a passenger in colorectal tumors, not a driver.
Subsequent research dismantled that objection methodically. Studies showed that F. nucleatum could accelerate colorectal tumor growth in animal models, that it suppressed anti-tumor immune responses, and that patients whose tumors contained high levels of the bacterium had worse outcomes and higher rates of recurrence. By 2020, the bacterium's role in colorectal cancer had moved from hypothesis to established science. It had become one of the clearest examples of a bacterium contributing directly to cancer progression.
But the colon made intuitive sense as a target. The gastrointestinal tract is teeming with bacteria, and the lining of the intestine is in constant contact with microbes. The breast, by contrast, was long considered essentially sterile. How could a mouth bacterium end up in breast tissue? And what would it do when it arrived?
From the Mouth to the Breast
The answer to the first question turns out to be surprisingly simple: the bloodstream. F. nucleatum enters the blood through inflamed, bleeding gum tissue, the hallmark of periodontal disease. Once in circulation, it can reach virtually any organ in the body. Researchers have detected the bacterium in placental tissue, in liver tumors, in pancreatic cancer, and now in breast tissue. The mouth, it turns out, is not a sealed compartment. It's a launch point.
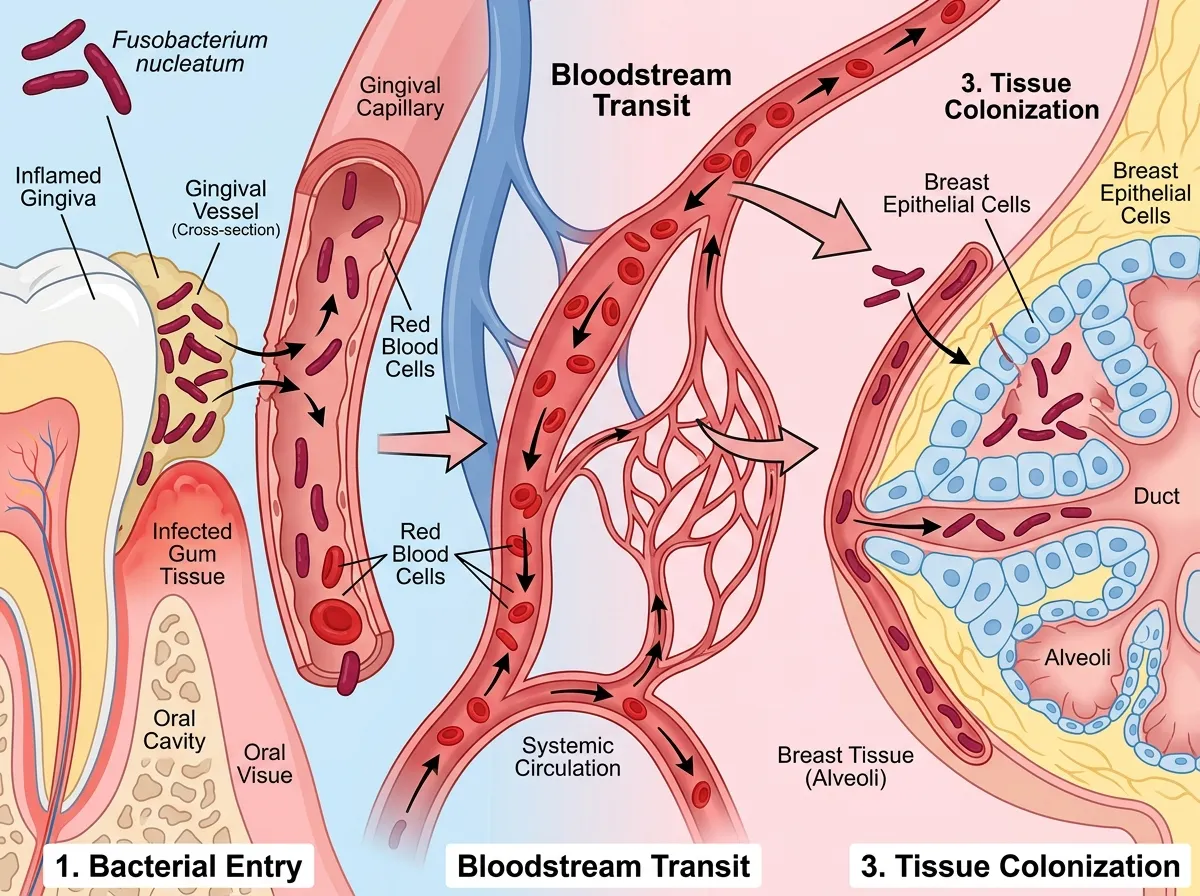
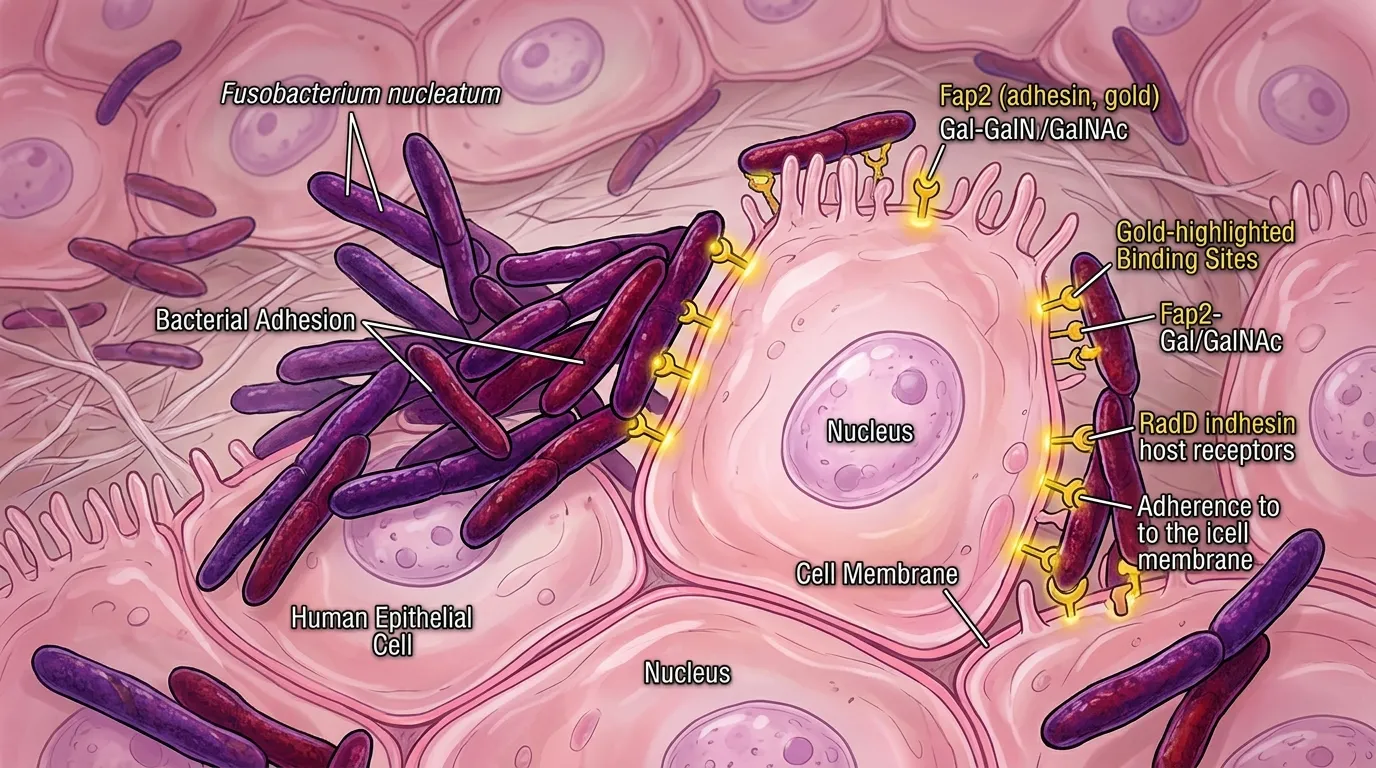
The Johns Hopkins team, led by Dipali Sharma, a professor of oncology at the Kimmel Cancer Center, didn't just confirm that F. nucleatum reaches breast tissue. They mapped what happens once it arrives. The bacterium binds to the surface of breast epithelial cells using a sugar molecule called Gal-GalNAc, a specific carbohydrate structure displayed on cell surfaces. This binding triggers the bacterium's entry into the cell, where the real damage begins.
What makes this sugar-mediated binding mechanism so significant is that it isn't random. Not all breast cells display the same amount of Gal-GalNAc on their surfaces. Cells with BRCA1 mutations, the same genetic variants that dramatically increase breast cancer risk through inherited susceptibility, express higher levels of this surface sugar. In practical terms, this means F. nucleatum has an easier time attaching to and entering the cells of people who are already genetically predisposed to breast cancer. The bacterium doesn't create vulnerability from nothing. It exploits a vulnerability that already exists, amplifying it in ways the genetic risk alone wouldn't predict.
The DNA Damage Cascade
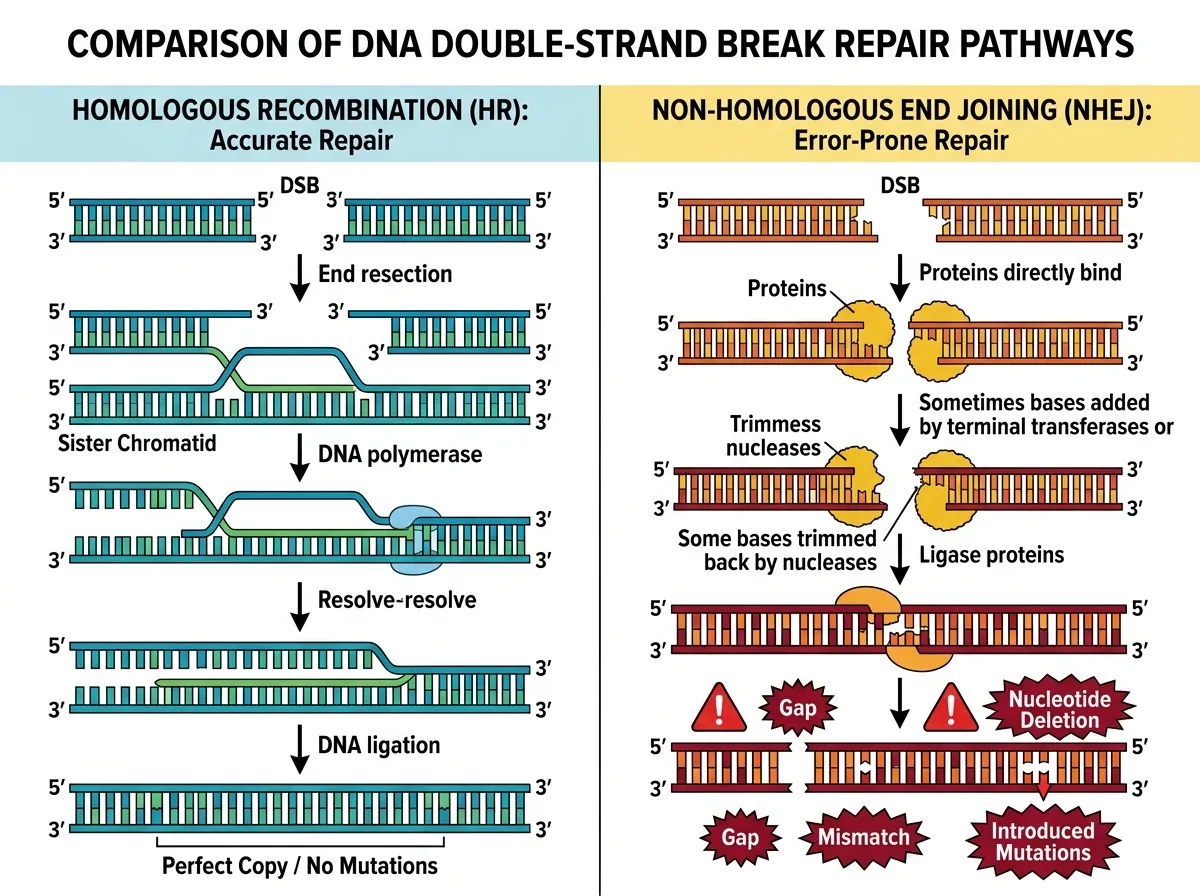
Once inside breast cells, F. nucleatum causes direct DNA damage. This alone isn't unique; many infections and environmental exposures damage DNA. What makes the bacterium's effect distinctive is what happens next: the type of repair pathway the cell activates in response.
Cells have multiple systems for fixing broken DNA. The most accurate is homologous recombination, which uses the undamaged copy of a gene as a template to reconstruct the broken strand precisely. But F. nucleatum exposure pushes cells toward a different system: nonhomologous end joining, or NHEJ. This repair pathway is fast but error-prone. It essentially glues broken DNA ends back together without checking whether the sequence is correct, introducing mutations in the process.
The shift toward error-prone repair is particularly dangerous in BRCA1-mutant cells, which already have compromised homologous recombination. BRCA1 protein is a key component of the accurate repair system. When it's dysfunctional, cells are already more reliant on backup pathways like NHEJ. Add F. nucleatum to the mix, and you get a compounding effect: more DNA damage arriving in cells that are less equipped to fix it properly, repaired by a system that introduces new mutations with each correction. The result is a rapid accumulation of genetic errors, exactly the kind of mutational burden that drives cells toward malignancy.
The research team documented this cascade in both cell culture experiments and animal models, showing that even brief exposure to F. nucleatum was sufficient to shift the DNA repair balance. This finding is important because it means the bacterium doesn't need to establish a permanent colony in breast tissue to cause lasting harm. A transient infection, cleared by the immune system within days, could still leave behind a legacy of improperly repaired DNA.
Beyond Mutation: How the Bacterium Changes Cell Behavior
DNA damage is only part of the story. The Johns Hopkins team found that F. nucleatum exposure also increases expression of a protein called DNA-PKcs (DNA-dependent protein kinase, catalytic subunit), a key enzyme in the NHEJ pathway. Elevated DNA-PKcs doesn't just mean more error-prone repair. It's associated with a cluster of behavioral changes in tumor cells that make cancer far more aggressive.
Cells with high DNA-PKcs expression showed enhanced migration, the ability to move through tissue rather than staying put. They demonstrated increased invasiveness, meaning they could penetrate basement membranes and enter new tissue compartments. They exhibited stem-like behavior, adopting characteristics of cancer stem cells that can seed new tumors. And they displayed increased resistance to chemotherapy, a property that makes established tumors harder to treat.
Dipali Sharma described the finding as revealing "a previously unrecognized environmental contributor to breast cancer that operates through specific, targetable molecular pathways." The specificity matters. If elevated DNA-PKcs is a measurable biomarker of bacterial-driven cancer progression, it could theoretically be used to identify patients whose tumors were influenced by F. nucleatum and to select treatments accordingly. DNA-PKcs inhibitors already exist in clinical development for other applications, raising the possibility that they could be repurposed for bacterially driven breast cancers.
In animal models, the consequences of these cellular changes were stark. Mice exposed to F. nucleatum developed breast tumors that grew faster, reached larger sizes, and, most alarmingly, spread to the lungs at significantly higher rates than tumors in unexposed control animals. The metastasis finding is particularly concerning because breast cancer mortality is driven overwhelmingly by metastatic disease, not by the primary tumor itself. If a common oral bacterium accelerates the transition from localized to metastatic cancer, it could help explain why some breast cancers progress rapidly while others remain indolent for years.
The Microbiome-Cancer Revolution
The F. nucleatum breast cancer connection doesn't exist in isolation. It's part of a larger shift in how researchers understand the relationship between the human microbiome and cancer, a shift that has gained extraordinary momentum over the past five years.
The story began with Helicobacter pylori, the stomach bacterium that causes ulcers and gastric cancer. When Barry Marshall and Robin Warren demonstrated this connection in the 1980s, it was considered radical. The idea that a chronic infection could cause cancer challenged the prevailing focus on genetics and environmental chemicals. Their Nobel Prize in 2005 validated the concept, but for years H. pylori was treated as an exception rather than a model.
It wasn't an exception. The list of microbiome-cancer connections has grown steadily. Certain strains of Escherichia coli produce a toxin called colibactin that directly damages DNA in colon cells. Bacteroides fragilis secretes a metalloprotease that promotes inflammation and colorectal tumor growth. Human papillomavirus and hepatitis B and C viruses drive cervical and liver cancers, respectively, through well-characterized mechanisms. The Epstein-Barr virus has been linked to multiple lymphomas and nasopharyngeal carcinoma. Each discovery reinforces the same principle: the organisms sharing our bodies are not passive bystanders. They are active participants in health and disease, including cancer.
What sets the F. nucleatum research apart from many of these examples is its cross-organ reach. Most microbiome-cancer connections are local: gut bacteria affecting gut cancer, skin bacteria affecting skin cancer, viral infections affecting the organ they infect. F. nucleatum starts in the mouth, travels through the blood, and causes cancer in the breast, a tissue with no direct microbial connection to the oral cavity. This long-range mechanism suggests that the microbiome's influence on cancer may extend far beyond the organs where bacteria physically reside. If mouth bacteria can drive breast cancer, what other distant connections remain undiscovered?
The implications for cancer prevention are significant, and they dovetail with a growing recognition that the body's systems are more interconnected than traditional medicine has acknowledged. Oral health, long siloed as a dental concern separate from medical care, may turn out to be a modifiable cancer risk factor. This doesn't mean that brushing your teeth prevents breast cancer, not directly, not yet. But it does mean that chronic periodontal disease, which affects nearly half of adults over 30 in the United States, provides a persistent source of bacteremia, bacteria entering the bloodstream, that could have consequences far from the mouth.
What This Means for BRCA1 Carriers
For the estimated one in 400 people who carry a BRCA1 mutation, the findings introduce a new variable into an already complicated risk calculus. BRCA1 carriers face lifetime breast cancer risks of 55 to 72 percent, depending on the specific mutation and family history. Current prevention strategies focus on enhanced screening (earlier and more frequent mammograms, breast MRI), risk-reducing medications (tamoxifen, aromatase inhibitors), and in some cases preventive surgery (bilateral mastectomy).
The F. nucleatum research suggests that oral health should be added to this conversation. BRCA1-mutant cells' elevated Gal-GalNAc expression creates a molecular welcome mat for the bacterium. This doesn't mean periodontal disease causes breast cancer in BRCA1 carriers, but it does mean that chronic gum infection may accelerate a process that genetics has already set in motion. The compounding of genetic susceptibility with bacterial exposure is a pattern that mirrors how daily habits accumulate into larger behavioral patterns without conscious awareness: small, repeated exposures building toward significant consequences.
Practically, this could mean that BRCA1 carriers should prioritize aggressive periodontal care as part of their cancer prevention strategy. Regular dental cleanings, prompt treatment of gum disease, and attention to oral hygiene take on a new dimension when the bacterium they control has been shown to specifically target cells with the same genetic vulnerability the patient carries. Whether insurance companies and clinical guidelines will incorporate this recommendation depends on confirmatory studies, but the biological logic is compelling.
The Bigger Picture
The Johns Hopkins findings sit at the intersection of microbiology, oncology, genetics, and preventive medicine, a convergence that would have been nearly impossible to imagine two decades ago, when these fields rarely overlapped. The research demonstrates that cancer risk is not simply a matter of genes or environment in isolation. It emerges from the interaction between the two, mediated by organisms we carry with us every day.
Several questions demand follow-up research. Can treating periodontal disease reduce breast cancer incidence in high-risk populations? Do antibiotics targeting F. nucleatum slow tumor progression in patients with established breast cancer? Are there other oral bacteria with similar long-range carcinogenic potential? And can the Gal-GalNAc binding mechanism be blocked pharmaceutically, cutting off the bacterium's access to vulnerable cells? Clinical trials exploring some of these questions are already in the planning stages at Johns Hopkins and other institutions.
The research also raises broader questions about the artificial boundaries in healthcare. Dental care and medical care operate in largely separate systems in most countries, with separate insurance, separate training, and separate clinical workflows. The discovery that a dental pathogen can drive breast cancer progression argues that this separation is not just inconvenient but potentially dangerous. A patient seeing an oncologist for BRCA1-related cancer screening might never be asked about her oral health, and her dentist might never know about her genetic risk. The biology doesn't respect these administrative boundaries.
For now, the practical takeaway is measured but real. F. nucleatum is not the sole cause of breast cancer, and good oral hygiene is not a guarantee against it. But the evidence that a treatable, preventable oral infection can accelerate breast cancer, particularly in genetically susceptible individuals, adds a new tool to the prevention toolkit. It's a tool that costs little, carries no side effects, and was sitting in the medicine cabinet all along.
Sources
- Oral Bacterium May Promote Breast Cancer Development and Spread - Johns Hopkins Medicine
- Gum disease bacteria linked to breast cancer development - ScienceDaily
- Gum disease bacteria may drive breast cancer - Science News
- Oral Fusobacterium nucleatum and Breast Cancer: Exploring a Microbial Contributor to Tumorigenesis - Pharmacy Times