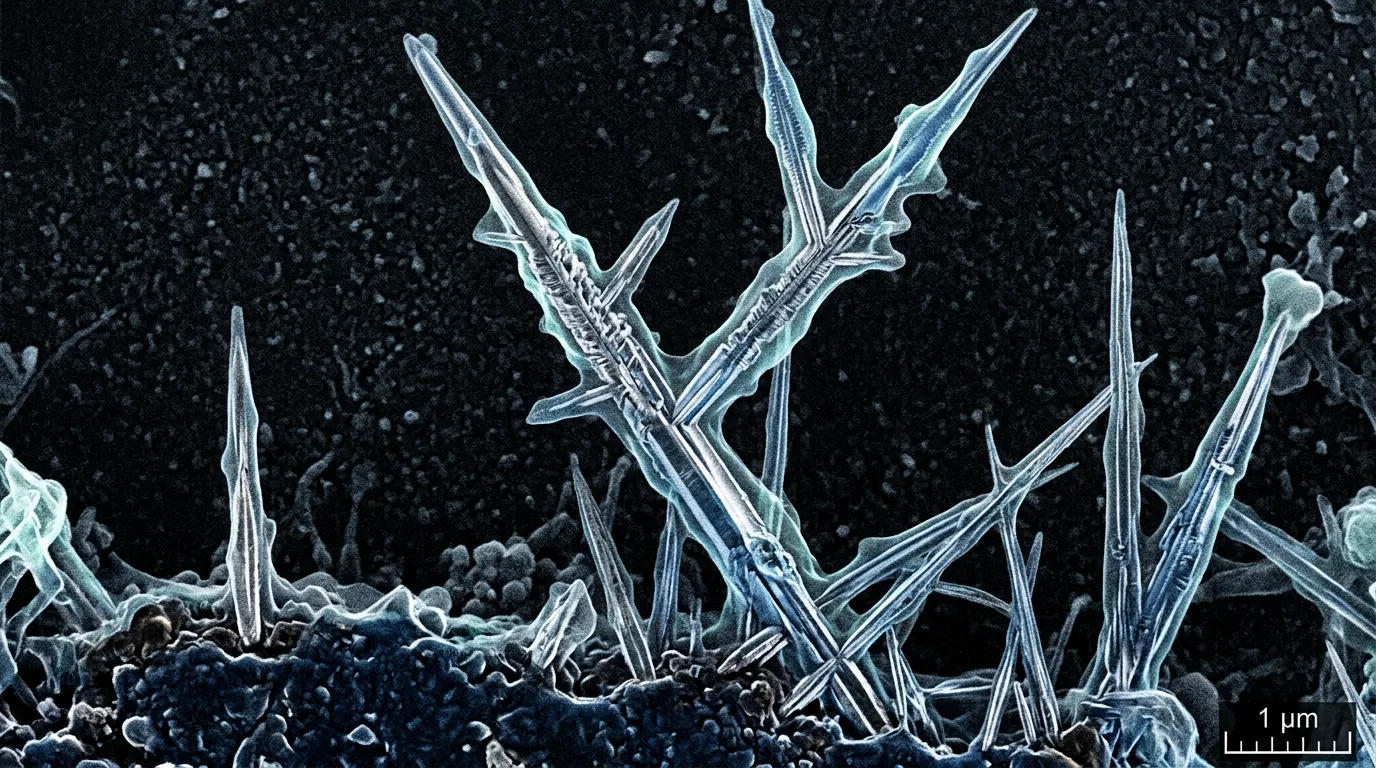
Every lithium-ion battery you own is growing microscopic metal needles inside itself right now. They form every time you charge your phone, your laptop, your electric toothbrush. For decades, scientists assumed these tiny structures, called dendrites, were soft and squishy, like the bulk lithium metal they grow from. That assumption turned out to be spectacularly wrong.
A team of researchers from Rice University, the New Jersey Institute of Technology, Georgia Tech, and several other institutions just published findings in Science that upend a core belief in battery research. Lithium dendrites aren't soft. They're strong, rigid, and brittle, snapping under stress like dry spaghetti rather than bending like Play-Doh. Their fracture strength exceeds 150 megapascals, roughly 250 times stronger than bulk lithium metal at 0.6 megapascals. To put that in perspective, these structures, thinner than a hundredth of a human hair, are mechanically closer to glass than to the metal they're made of.
The discovery matters because it explains, for the first time, why lithium batteries degrade, lose capacity, and occasionally catch fire in ways that existing models couldn't fully predict.
Needles in a Haystack of Assumptions
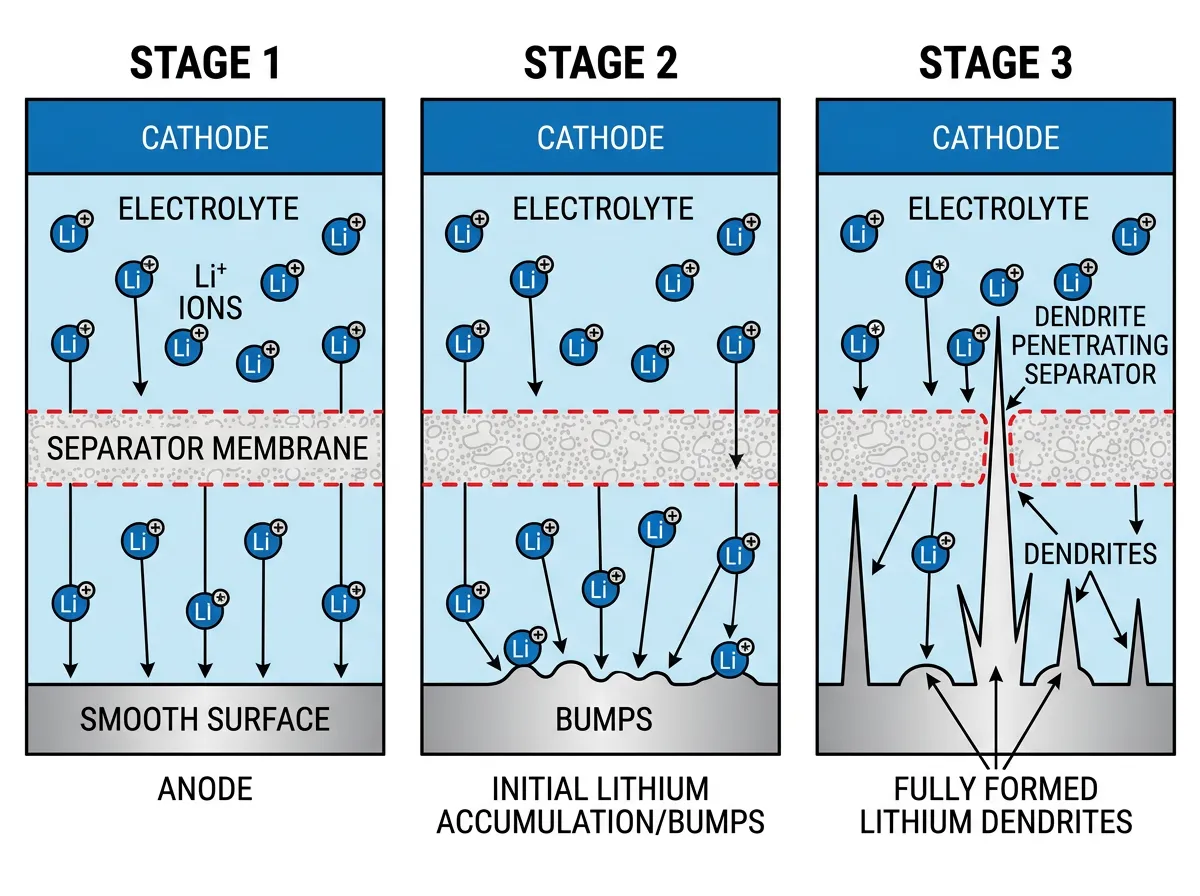
The dendrite problem has haunted battery engineers since lithium-ion cells first hit the market in 1991. During charging, lithium ions travel from the cathode to the anode and deposit as metallic lithium on the surface. Under ideal conditions, this deposition is smooth and uniform. Under real conditions, it isn't. Tiny irregularities on the anode surface create concentration points where lithium accumulates faster, sprouting into needle-like projections that grow with each charge cycle.
Battery researchers have known about dendrites for decades, and most strategies for dealing with them assumed they behaved like bulk lithium: soft, ductile, and deformable. The logic was straightforward. If dendrites are soft, then a sufficiently tough separator between the anode and cathode should stop them from puncturing through and causing a short circuit. Many separator designs and solid-state electrolyte formulations were built around this assumption. Some of those designs worked reasonably well, but nobody could fully explain why dendrites still managed to penetrate separators that should have been strong enough to contain them.
The problem, it turns out, was that nobody had actually measured the mechanical properties of individual dendrites. Lithium is extraordinarily reactive. It oxidizes almost instantly in air, and the dendrites themselves are nanoscale structures, about 100 times thinner than a human hair. Picking one up and testing it without destroying it or contaminating the sample is a technical nightmare.
How You Test Something You Can Barely See
Xing Liu, an assistant professor of mechanical and industrial engineering at NJIT and the study's co-lead author, described the core challenge in direct terms: "Lithium dendrites have long been assumed to be soft and ductile, like Play-Doh. But our observations suggest that they may instead be strong and brittle, snapping more like dry spaghetti."
Proving that required building an entirely new experimental workflow. The team grew dendrites on copper transmission electron microscopy (TEM) grids inside working lithium-ion battery cells with liquid electrolytes, mimicking real-world conditions as closely as possible. They then transferred the dendrite-laden grids to a nanomechanical testing device using an air-free procedure that prevented the lithium from reacting with moisture or oxygen. Every step, from growth to transfer to testing, had to happen inside carefully engineered protective environments.
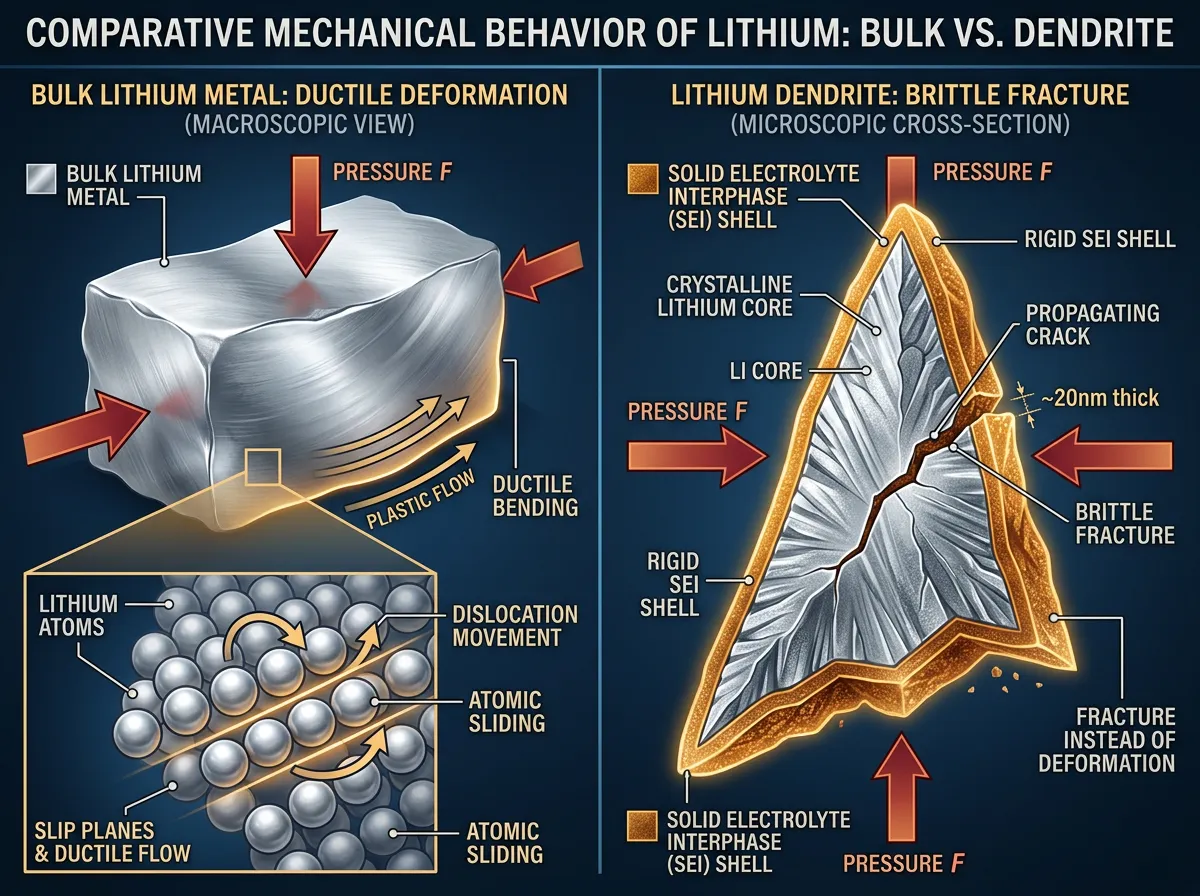
Jun Lou, Rice University's Karl F. Hasselmann Professor of Materials Science and Nanoengineering and the study's senior author, led the team at his Nanomaterials, Nanomechanics, and Nanodevices (N3) lab in directly probing individual dendrites. The researchers used cryo-transmission electron microscopy to image the internal structure of each dendrite at atomic resolution. What they found was a core-shell architecture: a single crystal of lithium metal at the center, wrapped in a roughly 15-nanometer-thick coating of solid electrolyte interphase (SEI), the chemical layer that forms naturally when lithium reacts with the surrounding electrolyte.
That SEI shell turned out to be the key to everything. Bulk lithium is soft because its atoms can slide past each other under stress, a property called ductility. But at the nanoscale, the rigid SEI coating constrains the lithium core so tightly that it can't deform. Instead of bending, the entire structure fractures catastrophically, like a glass rod rather than a copper wire.
A Fracture Problem, Not a Growth Problem
This distinction between soft and brittle matters enormously for how batteries fail. When a brittle dendrite pierces a battery separator and then fractures, it doesn't retreat harmlessly. The broken fragment becomes electrically disconnected from the anode, forming what battery scientists call "dead lithium," an inert metallic particle trapped inside the cell. Each fragment is lithium that can never participate in charging or discharging again. Over hundreds of charge cycles, these fragments accumulate. Your phone battery that once lasted all day and now dies by 3 PM? Dead lithium is a significant part of that story. If you've ever wondered why phone batteries degrade over time, dendrite fracture is one of the mechanisms driving that slow decline.
But capacity loss is the mild outcome. The severe one is thermal runaway. If a dendrite grows long enough to bridge the gap between anode and cathode before it fractures, it creates an internal short circuit. Current flows uncontrollably through the connection, generating heat, which accelerates chemical reactions inside the cell, which generates more heat. The result can be a fire or explosion. The U.S. Fire Administration documented over 1,000 lithium battery-related fires in 2022 alone, up from 800 in 2019. In the first months of 2025, more than a million lithium battery power banks were recalled by the Consumer Product Safety Commission after reports of overheating and fires.
The brittle-fracture discovery explains a pattern that previously puzzled engineers: why batteries sometimes fail suddenly after working fine for months. A ductile dendrite would grow slowly and predictably. A brittle one grows, hits a certain critical length, and snaps. The fracture redistributes stress and creates new nucleation points for fresh dendrite growth. It's a cascade mechanism, not a gradual decline. Liu noted the practical difficulty this creates: "At present, there is no practical method to 'clear' dendrites from a working battery cell."
The Nanoscale Size Effect: When Small Changes Everything
The most scientifically surprising element of this discovery is what it reveals about size-dependent material behavior, a phenomenon that extends well beyond batteries. Bulk lithium is one of the softest metals in existence. You can cut it with a kitchen knife. Yet at the nanoscale, constrained by a thin ceramic shell, it behaves like an entirely different material.
This is an example of what materials scientists call a size effect, and it appears across many domains. Gold, which is ductile and malleable at the macro scale, becomes extraordinarily strong in nanowire form. Silicon, brittle in bulk, can become flexible at nanoscale thicknesses. The same carbon atoms that form soft graphite also form diamond, depending on how they're arranged and constrained. The dendrite finding adds lithium to this growing catalog of materials that behave counterintuitively when you shrink them far enough.
What makes the lithium case particularly interesting is that the size effect isn't driven by the lithium itself. It's the SEI shell, a naturally forming chemical byproduct of battery operation, that transforms the mechanical properties. The researchers' simulations showed that without the SEI layer, lithium dendrites would indeed be soft and deformable. The coating is both the problem and the potential solution. If you could control the SEI layer's composition, thickness, or mechanical properties, you could potentially control whether dendrites are dangerous or benign.
This connects to a broader pattern in materials science that researchers studying exotic magnetic structures called skyrmions have also observed: nanoscale architectures often exhibit emergent properties that neither component possesses alone. The lithium core and the SEI shell are both unremarkable individually. Together, they produce a structure with mechanical properties that nobody predicted.

What This Means for the Next Generation of Batteries
The immediate practical implication is that separator design needs rethinking. If dendrites are brittle and strong rather than soft and weak, then making separators tougher may not be sufficient. You need separators that can absorb the force of a rigid needle without allowing penetration, a different engineering challenge than resisting the slow creep of a soft metal. Several research groups are already exploring ceramic-polymer composite separators that combine hardness with flexibility, but this study provides the mechanical data needed to optimize those designs with precision rather than guesswork.
The longer-term opportunity lies in controlling the SEI layer itself. Several groups, including a team at Brown University that published results in January 2026, are developing strategies to engineer artificial SEI layers with specific mechanical properties. If the natural SEI makes dendrites rigid and dangerous, an artificial alternative might keep them soft and harmless. Researchers at the Institute of High Performance Computing in Singapore, who collaborated on the Rice-NJIT study, used computational simulations to show that lithium alloy anodes could inhibit the formation of the problematic SEI coating, potentially eliminating the brittleness entirely.
This research also has implications for the solid-state battery race. Companies like Toyota, Samsung SDI, and QuantumScape have invested billions in solid-state batteries, which replace the liquid electrolyte with a solid material that is supposedly impervious to dendrite penetration. But dendrite formation remains the dominant failure mode in solid-state prototypes. Understanding that dendrites are rigid enough to exert over 150 megapascals of force explains why even solid electrolytes sometimes fail. The problem isn't that solid electrolytes are too weak; it's that dendrites are far stronger than anyone assumed.
For anyone following the parallel quest to harness fusion energy, there's an instructive analogy here. Both fields spent decades designing containment systems around incomplete models of what they were trying to contain. In fusion, the challenge is plasma instabilities that behave differently than simple models predicted. In batteries, it's dendrites that are mechanically different from what everyone assumed. In both cases, the path forward required first understanding the fundamental physics before engineering solutions could work.
The Deeper Question
The Rice-NJIT discovery is ultimately a story about the danger of untested assumptions in science. For over three decades, battery researchers designed around a model of dendrite behavior that had never been experimentally verified at the relevant scale. The assumption was reasonable, intuitive, and wrong. Bulk lithium is soft, so lithium dendrites must be soft. It took the development of new cryo-electron microscopy techniques and air-free nanomechanical testing methods to reveal the truth.
Liu and Lou's team hasn't solved the dendrite problem. What they've done is reframe it. The question is no longer "how do we stop soft metal from slowly pushing through separators?" It's "how do we prevent rigid, brittle needles from puncturing barriers and then fragmenting into capacity-killing dead lithium?" Those two questions lead to very different engineering approaches. The first leads to tougher barriers. The second leads to controlling the SEI chemistry that makes dendrites rigid in the first place, or to redesigning anode materials so dangerous dendrites never form.
Every lithium battery on Earth is still growing these tiny thorns with each charge cycle. Your phone, your laptop, the electric vehicle parked outside. The thorns are harder than anyone knew, and they're working against the battery's function every time you plug in. But for the first time, scientists can see exactly what they're dealing with, and that clarity is the first step toward batteries that don't slowly destroy themselves from the inside out.
Sources
- Why Do Lithium-ion Batteries Fail? Scientists Find Clues in Microscopic Metal 'Thorns' - NJIT News, March 2026
- New Lithium Battery Dendrite Strength Revealed - New Atlas, March 2026
- Strong and Brittle Lithium Dendrites - Science, 2026
- Designing with Hard, Brittle Lithium Needles Could Enhance Battery Safety - BioEngineer.org, 2026