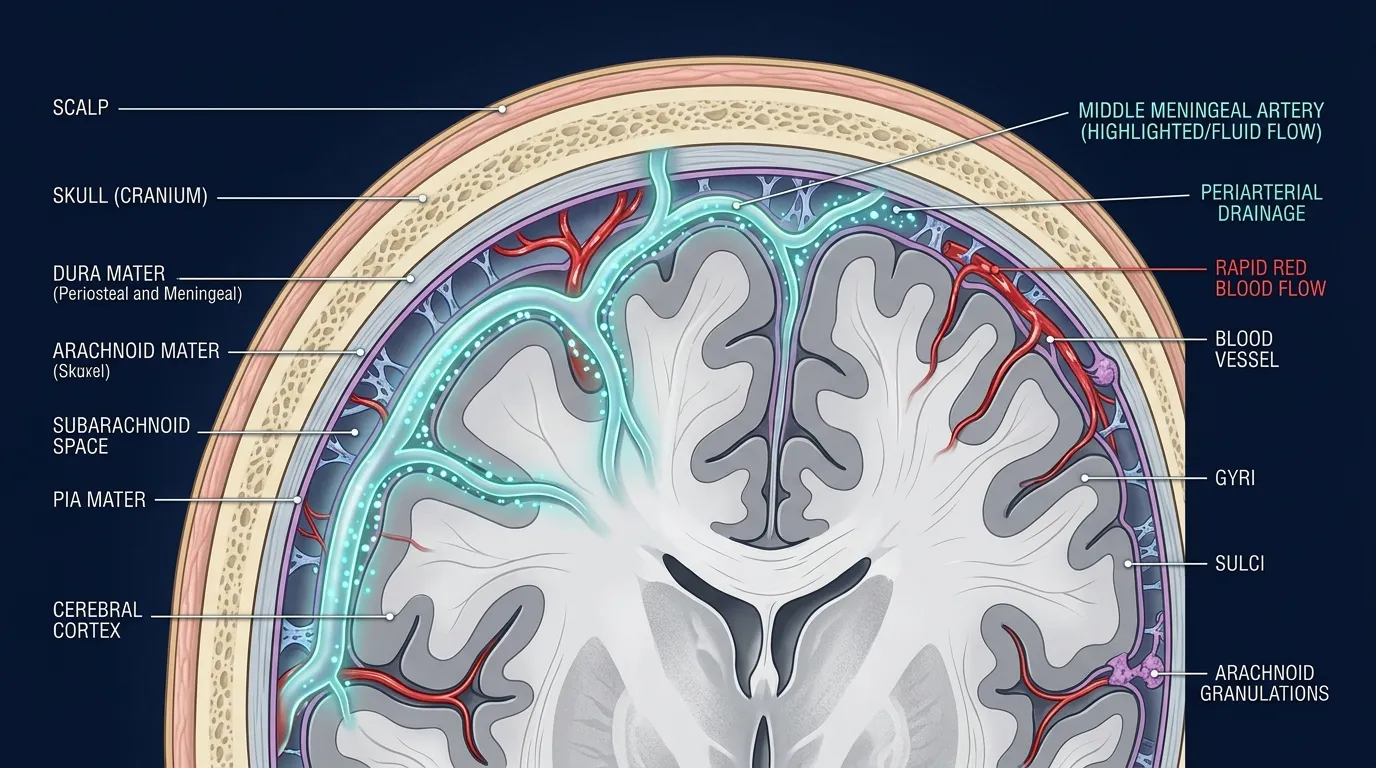
For most of the twentieth century, neuroanatomy textbooks described the middle meningeal artery with a kind of matter-of-fact boredom. It branches off the maxillary artery, passes up through the foramen spinosum, fans out between the dura and the skull, and feeds blood to the membranes that wrap the brain. Medical students memorize its path because it is the vessel most likely to rupture in an epidural hematoma after head trauma. Beyond that, the artery was considered a plumbing detail. It carried blood. It was not interesting.
A paper published in iScience last fall, and picked up this week across science news feeds after researchers shared new imaging, has just overturned that quiet consensus. A team led by Onder Albayram at the Medical University of South Carolina used a type of real-time MRI originally developed for NASA astronaut studies to track cerebrospinal and interstitial fluid around the middle meningeal artery in five healthy people. What they watched was not blood moving at the speed of blood. It was something slower, something that looked more like lymphatic drainage.
The finding adds a previously unmapped hub to the brain's waste-clearance network, the system responsible for pulling metabolic garbage out of neural tissue. It is the third significant rewrite of that anatomy in thirteen years, and the arc between the three discoveries is one of the most remarkable sequences in modern neuroscience.
The Old Dogma That Kept Breaking
For decades, medical students were taught that the brain does not have a lymphatic system. Every other organ in the body has lymph vessels, the low-pressure drainage network that clears interstitial fluid, waste products, and immune cells. The brain was different. The blood-brain barrier sealed it off. Whatever cleanup happened was presumed to diffuse out through the cerebrospinal fluid in ways no one had ever cleanly visualized. Textbooks published in the 2000s still listed the brain as lymph-free.
That claim was, in hindsight, a confession of ignorance dressed up as fact. The brain is the body's hungriest organ, consuming roughly 20% of its energy while holding only 2% of its mass. An organ that active produces waste. An organ that produces waste has to clear it somewhere. The textbook answer was "diffusion," which was less an answer than an admission that no one had bothered to look carefully.
When researchers finally did look, the maps changed fast.
The 2012 Moment: Nedergaard and the Glymphatic System
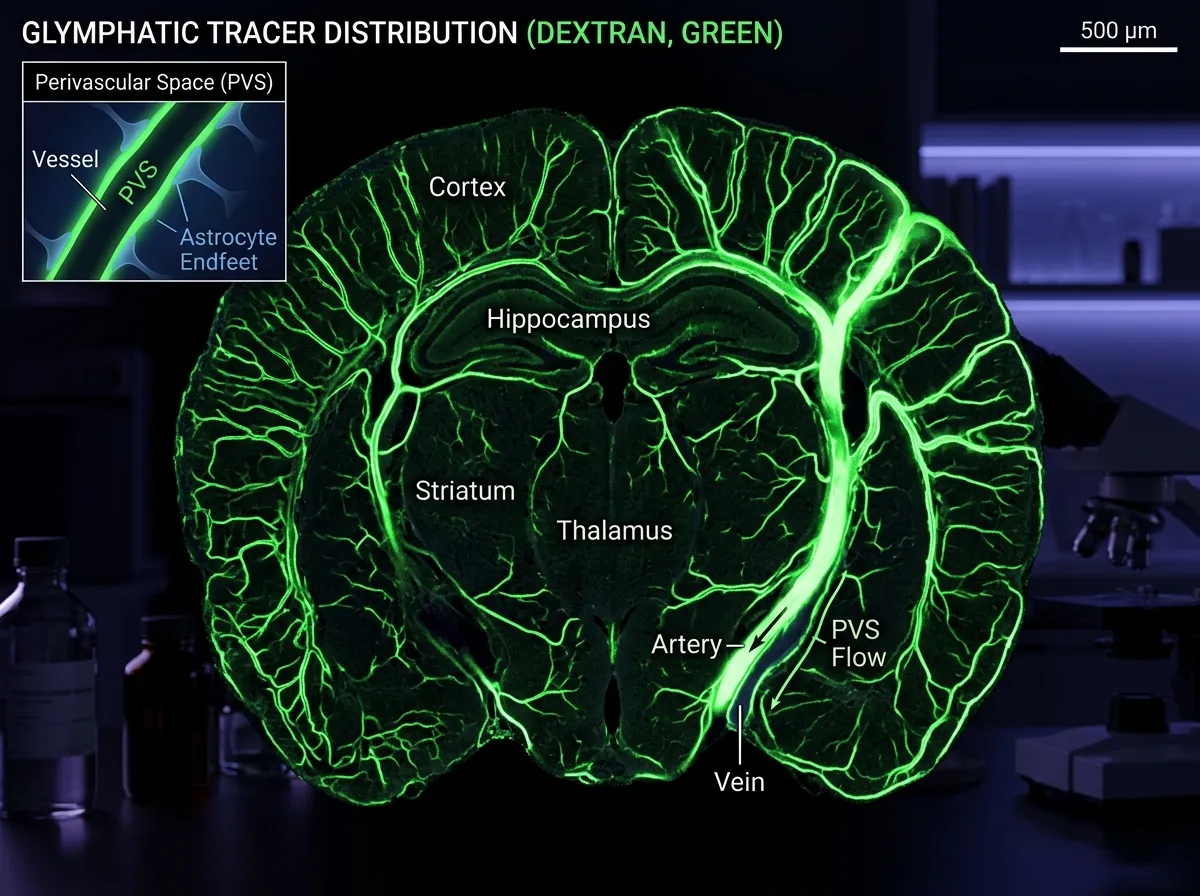
In 2012, Maiken Nedergaard, then at the University of Rochester, published a paper in Science Translational Medicine that did for brain drainage what the discovery of the circulatory system did for blood flow in the seventeenth century. Using two-photon microscopy to watch fluorescent tracers move through live mouse brains, her team showed that cerebrospinal fluid actively flows into the brain along the outside of arteries, mixes with interstitial fluid in the tissue, and then exits along veins, carrying waste proteins out with it.
Nedergaard called it the glymphatic system, a portmanteau of "glial" and "lymphatic," because the cells that enable the flow are glial cells called astrocytes. The aquaporin-4 water channels on those astrocytes act as the pumping machinery. The system is not a traditional lymphatic network of vessels with valves. It is a bulk-flow system that uses the space around blood vessels as its plumbing. That same astrocyte family turns out to be surprisingly active in other brain functions: a 2026 Boston University paper showed astrocytes store fear memories in ways previously thought to be the exclusive job of neurons, a finding that fits a broader pattern of glial cells being upgraded from support staff to active participants in brain function.
The second surprise came in 2013, when Nedergaard's group published a follow-up in Science. The glymphatic system works mostly when you are asleep. In sleeping mice, the interstitial space between brain cells expands by roughly 60%, and fluid washes through the tissue at rates more than ten times higher than in waking mice. Amyloid-beta, the protein that aggregates into Alzheimer's plaques, is one of the substances the system clears most efficiently during sleep. The old saying about needing sleep to "let your brain clean itself" turned out to be almost literally true.
For anyone following the Alzheimer's research world, that was a thunderclap. For the first time, there was a credible mechanistic story connecting sleep deprivation, amyloid buildup, and cognitive decline. If glymphatic flow slows with age, with poor sleep, or with disrupted circadian rhythm, waste accumulates. Waste accumulation is exactly what you see in the brains of Alzheimer's patients.
The 2015 Moment: Kipnis and the Lymphatic Vessels Everyone Missed
Three years later, in June 2015, Jonathan Kipnis and his team at the University of Virginia published a paper in Nature that did something even more surprising. They found actual lymphatic vessels inside the meninges, the protective membranes wrapping the brain. A separate group in Finland, led by Kari Alitalo, independently reported the same structures.
These were not the diffuse perivascular spaces Nedergaard had mapped. They were textbook lymphatic vessels: thin-walled tubes running through the dural sinuses, lined with the specific markers that lymphatic researchers had been using for decades to identify the system elsewhere in the body. They drained to cervical lymph nodes in the neck, exactly like lymphatic vessels from the rest of the body. And they had been sitting there, in every human cadaver ever dissected, waiting to be noticed.
The Kipnis finding blew open the dogma that the brain had no lymphatic system. It also reframed the entire field of neuroimmunology. If immune cells could move in and out of the brain through lymphatic vessels, then the brain was not the immune-privileged fortress it had been thought to be for a century. It was in constant, low-volume conversation with the rest of the body's immune system. That conversation had implications for multiple sclerosis, for brain tumors, for depression, and for every neurodegenerative disease that had ever been linked to inflammation.
Kipnis's lab later showed, in 2018, that meningeal lymphatic function declines with age in mice, and that boosting flow improves memory and reduces amyloid buildup. The glymphatic story and the lymphatic story, once viewed as competing frameworks, started to fit together as two parts of the same drainage network.
What Albayram's Team Just Added
The MUSC paper, "Meningeal lymphatic architecture and drainage dynamics surrounding the human middle meningeal artery," published in iScience under DOI 10.1016/j.isci.2025.113693, extends that map in a specific and important way. Everyone working on meningeal lymphatics had been focused on the dural sinus lymphatics Kipnis described. Albayram's group asked whether the middle meningeal artery, which runs through a different region of the dura, might also be a drainage hub. The anatomical reasoning was straightforward: wherever you have an artery in the dura, you also have the connective tissue environment in which lymphatic vessels like to form.
To test the idea, the team combined two kinds of evidence. First, they used dynamic contrast-enhanced MRI, a technique developed in collaboration with NASA to study how fluid shifts in astronauts' heads during spaceflight. The hardware had never been pointed at normal meningeal anatomy at this resolution before. They injected five healthy volunteers with a contrast agent and watched where it went over six hours. The fluid around the middle meningeal artery did something distinctive: it peaked at about 90 minutes and then cleared slowly, in a pattern characteristic of lymphatic drainage rather than arterial blood flow.
Second, they worked with Cornell University collaborators to perform immunofluorescence confocal microscopy and Hyperion Imaging Mass Cytometry on postmortem human tissue surrounding the artery. The tissue showed the molecular signatures of lymphatic endothelial cells, the specific cell type that lines lymph vessels. In plainer terms, the meninges around the middle meningeal artery contain structures built out of the same material as lymph vessels elsewhere in the body, arranged in patterns consistent with drainage function.
"We saw a flow pattern that didn't behave like blood moving through an artery," Albayram said in a statement released through EurekAlert. "It was slower, more like drainage, showing that this vessel is part of the brain's cleanup system."
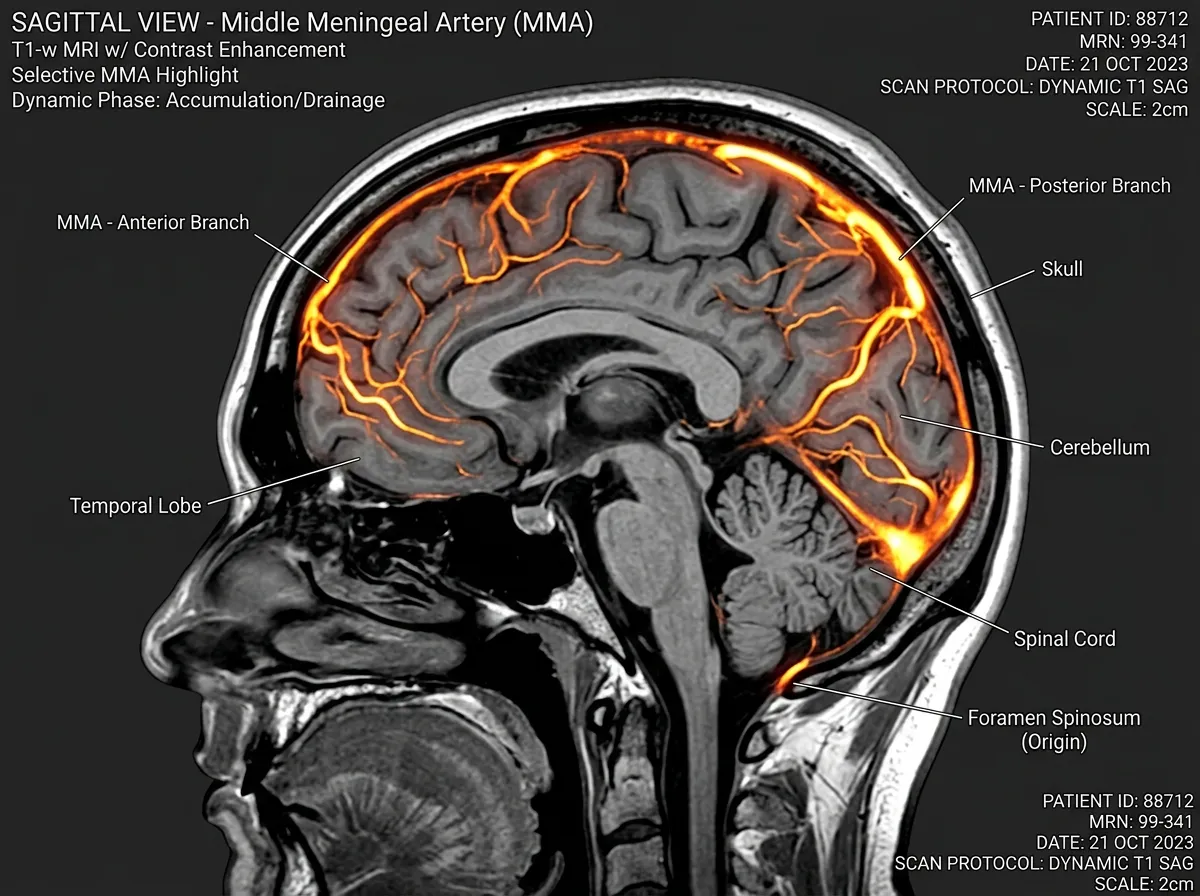
The study is small, which Albayram has been candid about. Five living participants is not a population. One postmortem tissue donor is not a comparative sample. What the paper provides is proof of concept, and proof of location, for a drainage site nobody had been looking at. Replicating the finding in larger samples, and watching what happens to it in aging or disease, is work that will take years.
Why the Location Actually Matters
One reason the middle meningeal artery is a promising drainage hub, rather than just another line on a map, is what sits near it. The artery runs along the inside of the temporal bone, close to the region where researchers have long observed accumulation of hyperphosphorylated tau protein in the brains of people who suffered repeated head impacts. That geographic overlap is why chronic traumatic encephalopathy (CTE), the degenerative condition now associated with football players, boxers, and veterans exposed to blast injuries, tends to show its earliest pathology in the temporal lobe.
If the drainage hub around the middle meningeal artery is damaged by repeated skull impacts, which is exactly the vessel most commonly torn in epidural bleeds, then one plausible story for CTE becomes: head trauma injures local lymphatic drainage, waste proteins accumulate in the tissue the damaged lymphatics used to clear, and neurodegeneration follows. That is a hypothesis, not a conclusion. But it is the kind of hypothesis you can only generate once you know the drainage hub is there to damage.
The same logic applies to Alzheimer's. The temporal lobe is among the earliest regions affected by Alzheimer's pathology, and the disease is increasingly understood as a failure of clearance as much as a failure of production. If local lymphatic drainage around the middle meningeal artery declines with age, amyloid and tau would accumulate fastest in exactly the regions where we observe Alzheimer's first striking. The idea that aging brains fail to clear specific molecules, rather than simply wearing out, has been picking up experimental support from multiple directions. UCSF researchers recently identified FTL1 as a single protein whose reduction can reverse cognitive decline in aged mice, suggesting that accumulation-and-clearance frameworks may be the dominant story for how brains age.
The Sleep Question Nobody Has Answered Yet
Nedergaard's 2013 finding that glymphatic flow is a sleep-dependent phenomenon is probably the single most cited piece of neuroanatomy research of the past decade. It has been used to explain why chronic sleep deprivation correlates with elevated amyloid levels in cerebrospinal fluid, why night-shift workers show higher rates of cognitive decline, and why sleep apnea is a risk factor for Alzheimer's. Whether the newly identified middle meningeal drainage hub is similarly sleep-dependent is, at the moment, an open question.
There are reasons to suspect it might be. The glymphatic and meningeal lymphatic systems appear to function as connected stages of the same pipeline: glymphatic flow pushes waste out of the brain parenchyma into the cerebrospinal fluid, where meningeal lymphatics then carry it away to the neck. If the upstream part of the pipeline is sleep-dependent, the downstream part probably has to keep pace. But nobody has yet run the experiment of scanning drainage at the middle meningeal artery in sleeping versus waking subjects, in part because the techniques required to do so at adequate resolution only just became possible.
That experiment is the obvious next step, and the one most likely to generate another headline-grade result. If the MMA drainage hub turns out to be sleep-dependent in humans the way the glymphatic system is in mice, the list of behaviors we know affect brain waste clearance, sleep quality, sleep duration, circadian alignment, sleep position, suddenly has a new anatomical target to trace them through.

The Bigger Picture
The sequence matters: 2012, 2015, 2025. Three discoveries, each one arriving roughly when the previous one had enough momentum to suggest where to look next. Nedergaard's glymphatic finding told the field that bulk fluid flow through brain tissue was real. Kipnis's meningeal lymphatic finding told the field that the fluid had real lymphatic vessels to drain into. Albayram's middle meningeal artery finding tells the field that those vessels are in more places than anyone had mapped, including exactly the place where head injuries most commonly cause visible bleeding.
None of these three findings would have been easy to make in a vacuum. Each one depended on a specific technical advance: two-photon microscopy for Nedergaard, immunohistochemistry with lymphatic-specific markers for Kipnis, dynamic contrast MRI adapted from NASA spaceflight research for Albayram. None of those techniques existed in their current form a decade before the discovery they enabled. The reason the brain drainage maps had been wrong for so long was not that anyone was lazy. It was that nobody could see well enough to notice the pipes. "We do not know" and "it is not there" are very different claims, and the history of neuroscience is thick with mistakes that collapsed the first into the second.
What the Albayram paper adds is not a cure for Alzheimer's or a fix for CTE. It is something more preliminary and more fundamental: a new place on the map, and a new site at which to ask old questions. Why does brain waste clearance slow with age? Where, specifically, does it break? Which structures are damaged first by head injury, by sleep disruption, by vascular disease? What can you do to keep the drainage pipeline working longer?
Those questions were already in play before the middle meningeal artery finding. What the finding does is sharpen them, by giving researchers one more hub to measure, to perturb, and to protect. The glymphatic system revealed that the brain has a cleanup process tied to sleep. The meningeal lymphatics revealed that the cleanup has anatomical plumbing. The MMA drainage hub reveals that the plumbing extends to regions with specific clinical relevance.
Taken together, the three discoveries are a quiet argument that much of what is interesting about how the brain ages, how it fails, and how it maintains itself is happening in the fluid movements we were not looking at for most of the history of neuroscience. Thirteen years is not a long time to have reorganized the field. The rate at which the maps are still being rewritten suggests there is more to come, and probably sooner than anyone expects.
"A major challenge in brain research," Albayram said in the EurekAlert statement, "is that we still don't fully understand how a healthy brain functions and ages. Once we understand what 'normal' looks like, we can recognize early signs of disease and design better treatments." The middle meningeal artery, long ignored, is now part of what normal looks like.
Sources
- Meningeal lymphatic architecture and drainage dynamics surrounding the human middle meningeal artery, Albayram et al., iScience Vol. 28, Issue 11 (November 21, 2025)
- Scientists just found a hidden "drain" inside the human brain, ScienceDaily, April 8, 2026
- Unlocking the brain's hidden drainage system, EurekAlert, April 2026
- Structural and functional features of central nervous system lymphatic vessels, Louveau, Kipnis et al., Nature, 2015
- Glymphatic system: A Beginner's Guide, PMC review article on Nedergaard's foundational work