Among the thousands of proteins circulating in a mouse brain, researchers at the University of California, San Francisco wanted to find the ones that changed most as the brain aged. They tracked shifts in gene expression and protein levels across the hippocampus, the region most responsible for forming and retrieving memories, in mice of different ages. Out of everything they measured, one protein stood out with unusual consistency. It was present at low levels in young animals and elevated levels in old ones, and every time it climbed, brain function declined.
That protein is called FTL1, short for ferritin light chain 1. It belongs to the ferritin family, a group of proteins best known for storing iron inside cells. But its role in the aging brain turns out to be far more interesting than iron management. When the team artificially raised FTL1 in young mice, their brains started behaving like old ones. When they lowered it in old mice, something remarkable happened: the animals' brains started recovering. Connections between neurons rebuilt. Memory performance improved. The decline wasn't just slowed. It reversed.
"It is truly a reversal of impairments," said Saul Villeda, PhD, associate director of the UCSF Bakar Aging Research Institute and the study's senior author. "It's much more than merely delaying or preventing symptoms."
The Search for a Single Culprit
The study, published in Nature Aging, began with a deceptively simple question: what molecular changes in the hippocampus track most reliably with aging? The hippocampus is one of the first brain regions to deteriorate with age, and its decline shows up as the kind of everyday memory problems that most people over 60 recognize, forgetting names, misplacing objects, struggling to recall recent conversations.
Villeda's team took a broad approach. Rather than testing a hypothesis about a single pathway, they screened the entire proteome of the hippocampus across multiple age groups of mice, looking for proteins whose levels shifted consistently between youth and old age. Most proteins showed noisy, inconsistent patterns. FTL1 was different. Its levels rose steadily as animals aged, and the increase correlated tightly with two measurable outcomes: fewer synaptic connections between neurons and worse performance on memory tests.
The consistency was striking. In neuroscience, single-protein explanations for complex processes like aging are rare. The brain's decline typically involves cascading failures across multiple systems, inflammation, oxidative stress, vascular damage, protein aggregation. Finding one molecule that tracked so cleanly with cognitive decline was unexpected, and it raised an obvious follow-up question: was FTL1 a bystander, simply rising alongside other aging processes, or was it actually driving the damage?

What FTL1 Does to an Aging Brain
To test whether FTL1 was a cause rather than a symptom, the team ran two complementary experiments. First, they used genetic tools to boost FTL1 levels in the hippocampus of young, healthy mice. The results were immediate and clear. The young mice's neurons developed simplified structures, losing the complex branching patterns that allow brain cells to form rich networks of connections. Their performance on standard memory tasks dropped to levels normally seen in much older animals. Artificially aging one protein had, in effect, artificially aged the brain.
The mechanism appears to involve cellular energy production. Higher FTL1 levels suppressed metabolic activity in hippocampal cells, slowing the energy-intensive processes that neurons depend on to maintain their connections and fire properly. When the researchers treated these cells with a compound that stimulates metabolism, the harmful effects of elevated FTL1 were prevented. The protein wasn't damaging neurons directly. It was starving them of the energy they needed to stay healthy.
This metabolic connection adds an important dimension to our understanding of brain aging. Neuroscientists have known for years that the brain's energy systems decline with age, but the upstream causes of that decline have been difficult to pin down. FTL1 now appears to be one specific trigger, a protein whose accumulation gradually throttles the metabolic machinery that keeps neurons connected and functional.
The Reversal That Changed the Conversation
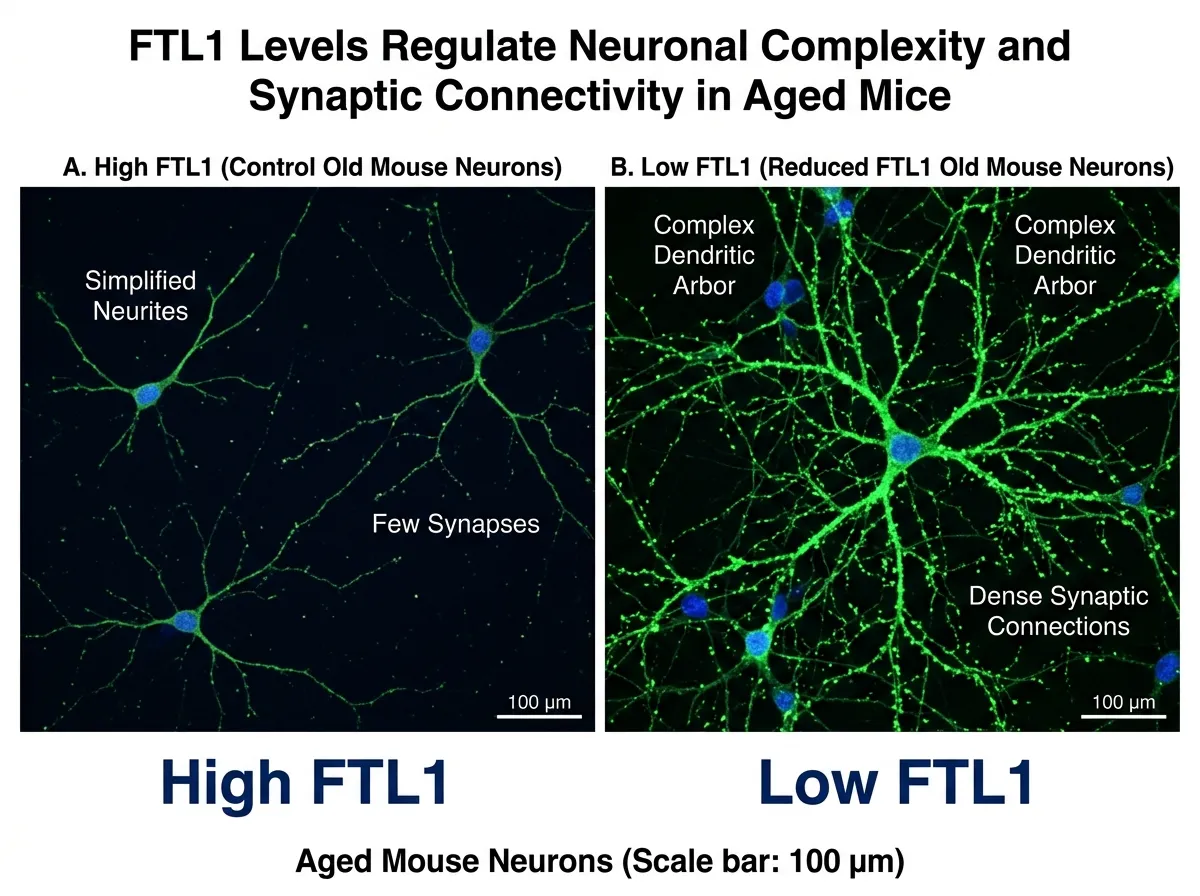
The most striking results came from the second experiment. The team used the same genetic tools to reduce FTL1 levels in old mice, animals that had already developed the memory problems and connectivity losses associated with aging. If FTL1 was only part of a larger cascade, lowering it in already-impaired animals might have little effect. The damage, in theory, could be done.
That isn't what happened. Old mice with reduced FTL1 showed increased synaptic connections in their hippocampus. Their neurons regained some of the complex branching architecture they had lost. And on memory tests, the animals performed measurably better than untreated old mice, approaching levels that looked more like middle-aged animals than elderly ones.
The word "reversal" is used cautiously in aging research, and for good reason. Most interventions in the field manage to slow decline or prevent it from worsening, not undo damage that has already occurred. Villeda himself emphasized the distinction: this wasn't prevention, it was recovery. The old mice's brains didn't just stop deteriorating. They rebuilt, at least partially, what they had lost.
Iron, Ferritin, and a Decades-Old Mystery
FTL1's identity as a member of the ferritin family adds an intriguing layer to these findings. Ferritin proteins are the body's primary mechanism for storing iron safely inside cells, preventing the metal from generating the kind of reactive oxygen species that damage DNA, lipids, and proteins. Iron accumulation in the brain has been observed in aging and in neurodegenerative diseases including Alzheimer's and Parkinson's for decades, but the relationship between that iron buildup and cognitive decline has remained frustratingly unclear.
Some researchers have proposed that excess iron directly damages brain tissue through oxidative stress. Others have argued that iron accumulation is a secondary effect of other aging processes, a consequence rather than a cause. The FTL1 findings suggest a third possibility: that the ferritin proteins the brain produces to manage its rising iron levels may themselves be part of the problem. FTL1 doesn't appear to harm neurons through iron-related oxidative damage. Instead, it disrupts their metabolism, creating a bottleneck in cellular energy production that weakens synaptic connections over time.
This reframing could redirect research into brain protein dysfunction and neurodegeneration. If FTL1's harmful effects come from metabolic suppression rather than iron toxicity, then the therapeutic target isn't removing iron from the brain, an approach that has produced disappointing clinical results, but rather blocking or reducing FTL1 specifically while leaving the rest of the iron management system intact.
From Mice to Medicine
The gap between a mouse study and a human therapy is wide, and Villeda's team is careful not to overstate what their findings mean for clinical practice today. Mice are not humans, the mouse hippocampus is not the human hippocampus, and a genetic manipulation that works cleanly in a controlled laboratory setting may not translate to a drug that works safely in a living person.
That said, several features of this research make it more promising than the average mouse aging study. The fact that FTL1 was identified through an unbiased screen, rather than selected based on a pre-existing theory, reduces the risk that the finding reflects researcher bias. The bidirectional results, raising FTL1 causes decline while lowering it reverses decline, provide stronger causal evidence than a single-direction experiment would. And the metabolic mechanism offers a concrete target for drug development, since compounds that modulate cellular metabolism are already well-studied in other contexts.
The study was funded by the National Institutes of Health, the Simons Foundation, the Bakar Family Foundation, and several other institutions that have invested heavily in translational aging research. Whether FTL1 itself becomes a drug target or whether it points researchers toward a broader metabolic pathway that can be modulated, the finding opens a new line of investigation that didn't exist before this work was published.

Where This Leads
The FTL1 discovery sits at an intersection that neuroscience has been trying to reach for years: a specific, targetable molecule that appears to drive a significant portion of age-related cognitive decline. The field is littered with past candidates that looked promising in mice and failed in humans, and healthy skepticism is warranted. But the strength of this study lies in its specificity. Rather than identifying a broad process like "inflammation" or "oxidative stress," it points to one protein, one mechanism, and one measurable outcome.
The open questions are substantial. Does FTL1 accumulate in the human hippocampus the same way it does in mice? Does reducing it in older humans produce the same recovery of synaptic connections? Are there side effects to lowering a protein that presumably serves some functional role in iron storage? And perhaps most importantly, does FTL1 interact with the protein aggregation pathways, tau tangles and amyloid plaques, that define Alzheimer's disease, or does it operate through an entirely separate mechanism?
Those questions will take years to answer. What the UCSF team has provided is not a treatment but a direction, a single clear thread in a field that has been searching for one. If the thread holds, it could reshape how we think about the aging brain: not as a system undergoing inevitable, irreversible decline, but as one being throttled by a specific molecular brake that, at least in mice, can be released.
Sources
- Scientists found a protein that drives brain aging, and how to stop it - ScienceDaily
- This Protein Slows the Aging Brain, and We Know How to Counter It - UCSF News
- Targeting iron-associated protein Ftl1 in the brain of old mice improves age-related cognitive impairment - Nature Aging
- Can you turn off brain aging? New study suggests it might be possible - Deseret News