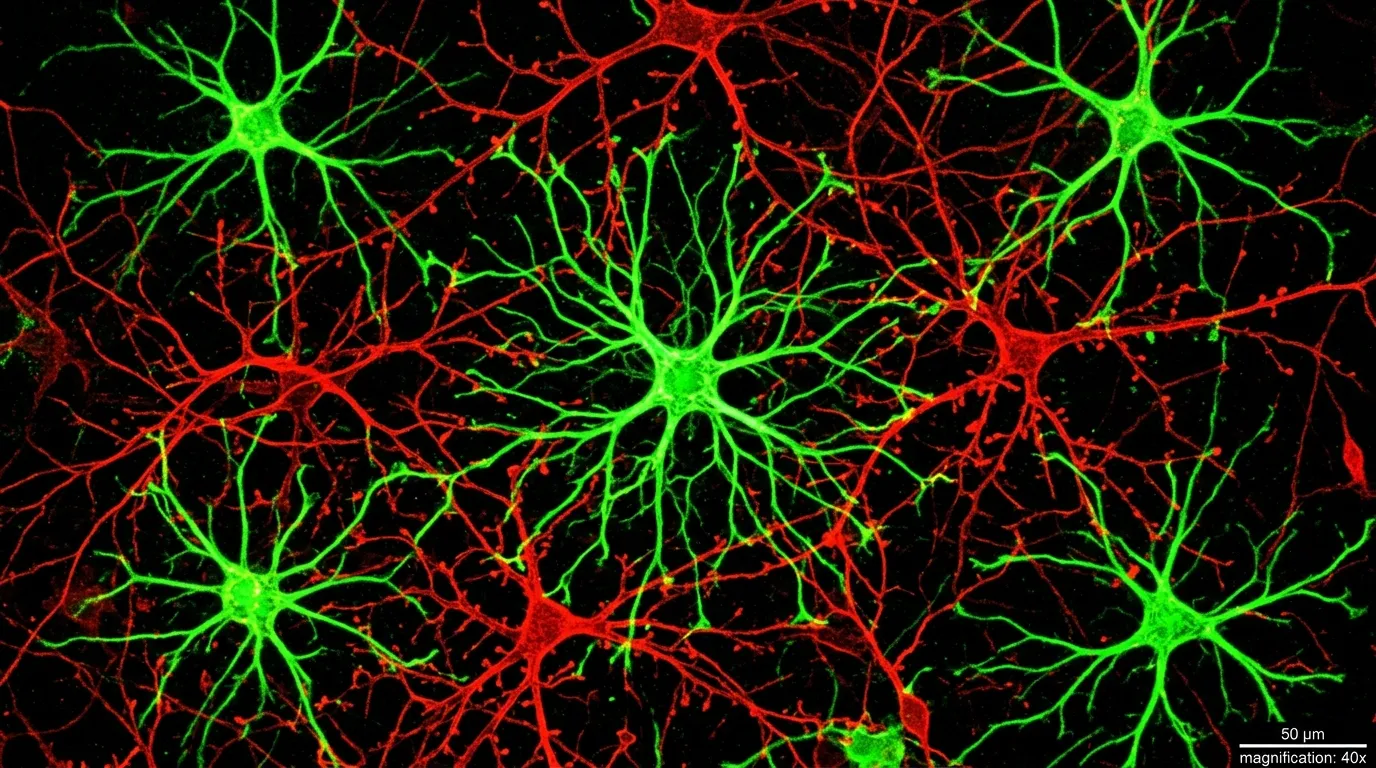
For most of the history of neuroscience, the brain had a clear hierarchy. Neurons did the thinking. Everything else was support staff. Astrocytes, the star-shaped cells that outnumber neurons in many brain regions, were filed under "structural maintenance": they cleaned up chemical waste, delivered nutrients, and formed the scaffolding that held the real workers in place. Calling them glial cells, from the Greek word for "glue," captured exactly how seriously researchers took them.
That framework just collapsed. A multi-institutional study published in Nature, led by researchers at the National Institutes of Health, has demonstrated that astrocytes in the amygdala don't just support fear memory processing. They actively encode fear signals, amplify them during recall, and quiet them during extinction. Remove astrocytes from the equation, and neurons can't form proper fear responses at all. The discovery doesn't tweak our model of how the brain handles fear. It rebuilds the model from the foundation up, with direct implications for how we treat conditions like PTSD, anxiety disorders, and phobias.
A Century of Being Overlooked
The neuron doctrine, the idea that nerve cells are the fundamental signaling units of the brain, dates to Santiago Ramón y Cajal's work in the 1890s. Cajal won the Nobel Prize for it in 1906, and the framework proved so productive that it dominated neuroscience for over a century. Researchers mapped neural circuits for vision, movement, language, and emotion, building an increasingly detailed wiring diagram of the brain with neurons as the only components that mattered.
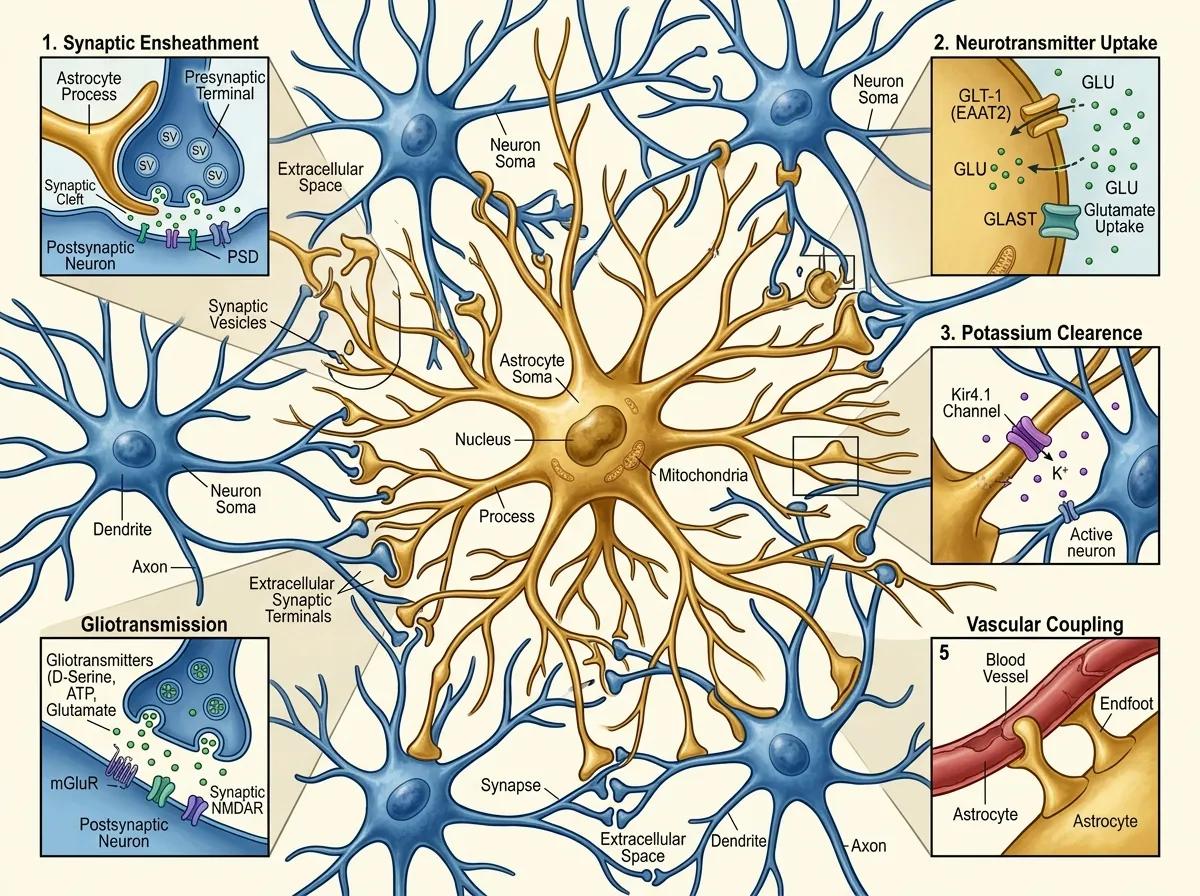
Astrocytes barely appeared in that diagram. They were known to regulate blood flow to active brain regions, maintain the chemical environment around synapses, and help form the blood-brain barrier. Important work, but fundamentally custodial. Even as evidence accumulated through the 2000s and 2010s that astrocytes could respond to neurotransmitters and release signaling molecules of their own, the dominant view held that these were modulatory effects, fine-tuning the real work that neurons performed.
The NIH team, led by Andrew Holmes and Olena Bukalo at the Laboratory of Behavioral and Genomic Neuroscience, suspected the division of labor was far less clean than textbooks suggested. Their focus was the amygdala, the almond-shaped structure deep in the temporal lobe that serves as the brain's threat detection center. If astrocytes were doing more than housekeeping anywhere in the brain, this was the place to look.
What the Study Actually Found
The research team used mouse models fitted with fluorescent activity sensors that could track astrocyte behavior in real time. The sensors lit up when astrocytes became active, allowing researchers to watch these cells respond as fear memories formed, were recalled, and gradually faded through a process called extinction.
The results were unambiguous. During fear conditioning, when mice learned to associate a neutral tone with a mild shock, astrocyte activity in the amygdala surged alongside neuronal activity. This alone was notable: the cells weren't just present during fear learning. They were actively participating. But the more striking finding came during recall. When mice later heard the tone without the shock, astrocyte activity increased again, tracking closely with the strength of the animal's fear response.
"For the first time, we found that astrocytes encode and maintain neural fear signaling," said Lindsay Halladay, an assistant professor of neuroscience at the University of Arizona and a senior author on the study.
The team then pushed further. Using targeted techniques to increase or suppress astrocyte signaling, they demonstrated a direct causal relationship between astrocyte activity and fear memory strength. When astrocyte signals were amplified, fear responses intensified. When they were dampened, fear responses weakened. And when astrocyte function was disrupted entirely, neurons in the amygdala could not form the coordinated activity patterns necessary for normal fear processing.
Perhaps most importantly for clinical applications, the researchers found that astrocyte activity declined naturally as fear memories were extinguished through repeated exposure to the conditioned stimulus without the shock. This mirrors the mechanism behind exposure therapy, the gold-standard treatment for PTSD and anxiety disorders. The finding suggests that successful fear extinction depends not just on neuronal relearning, but on astrocytes actively releasing their grip on the fear signal.
The Ripple Effect Beyond the Amygdala
One of the study's most intriguing findings was that disrupting astrocyte activity in the amygdala produced effects that extended to the prefrontal cortex, the brain region responsible for executive decision-making. When amygdala astrocytes were suppressed, prefrontal neurons showed altered activity patterns during fear-related tasks, suggesting that astrocytes in one brain region can influence neural processing in distant regions.
This finding resonates with a pattern that has been emerging across several recent neuroscience discoveries. The brain's signaling architecture is far more distributed and interconnected than the neuron-centric model predicted. Research on brain proteins involved in neurodegeneration has similarly revealed that non-neuronal cells play active roles in processes once attributed entirely to neurons. And studies on how the brain manages memory deletion have shown that forgetting is an active, regulated process, not a passive decay, a framework that the astrocyte findings reinforce from a completely different angle.
The cross-regional signaling also raises questions about astrocyte involvement in other emotional and cognitive processes. If these cells co-process fear in the amygdala, do they play analogous roles in reward processing in the nucleus accumbens, or spatial memory in the hippocampus? The study doesn't answer those questions directly, but the methodology it establishes provides a template for investigating them.

What This Means for Treating PTSD
Post-traumatic stress disorder affects roughly 6% of the U.S. population at some point in their lives, with rates substantially higher among combat veterans and survivors of sexual assault. The condition is defined by fear memories that refuse to extinguish: a car backfire triggers a combat flashback, a particular smell resurrects a traumatic event, and the brain's threat detection system stays locked in a state of perpetual alarm.
Current treatments work, but imperfectly. Exposure therapy, which gradually reduces fear responses through controlled re-exposure to triggering stimuli, succeeds in about 50 to 60% of PTSD patients. SSRIs, the most commonly prescribed medication, help roughly the same proportion. Both approaches target neuronal pathways. Neither was designed with astrocytes in mind.
The NIH findings suggest that treatment-resistant PTSD may involve astrocytes that continue to maintain fear signals even as neurons begin the extinction process. If astrocyte activity is the bottleneck preventing successful fear extinction, then therapies targeting astrocyte-specific pathways could complement existing neuron-focused treatments, potentially reaching patients who don't respond to current options.
This is not a speculative leap. The study demonstrated that manipulating astrocyte activity directly alters fear memory strength in a controlled experimental setting. The challenge now is translating that mechanism into clinically viable interventions. Astrocytes respond to different chemical signals than neurons, which means existing psychiatric drugs, most of which were designed to modulate neurotransmitter systems, may not be the right tools. New pharmacological approaches targeting astrocyte-specific receptors and signaling pathways would need to be developed and tested, a process that typically takes years but now has a clear biological rationale to support it.
A Deeper Pattern in Modern Neuroscience
The astrocyte discovery belongs to a broader shift in how brain science understands complexity. For decades, the field operated under an assumption that could be loosely summarized as "find the right neuron, find the answer." Map the circuit, identify which neurons fire during which behavior, and the mechanism reveals itself. This approach produced enormous progress, from deep brain stimulation for Parkinson's disease to our understanding of how visual cortex processes edges and shapes.
But the neuron-only framework also left conspicuous gaps. Fear conditioning was one of the most thoroughly studied phenomena in all of neuroscience, with decades of research mapping the amygdala circuits involved. Yet the role of the most abundant cell type in those circuits went unexamined. The fact that astrocytes could hide in plain sight for this long, in one of the best-studied brain regions, for one of the best-studied behavioral phenomena, suggests that other fundamental discoveries may be waiting in similarly well-trodden territory.
The parallel to genetics is instructive. For years after the Human Genome Project, researchers focused on protein-coding genes, the 1.5% of DNA that produces proteins. The remaining 98.5% was dismissed as "junk DNA." It turned out to contain regulatory elements that control when and how genes are expressed, fundamentally reshaping our understanding of heredity and disease. Astrocytes may be the brain's version of junk DNA: abundant, conspicuous, and dismissed precisely because they didn't fit the dominant framework.
The Bigger Picture
The most significant aspect of this research may not be what it tells us about fear, but what it reveals about the limits of our previous methods. When neuroscientists had tools that could only measure neuronal activity, they built models that only included neurons. The development of fluorescent sensors capable of tracking astrocyte behavior in real time didn't just produce a new finding. It exposed a blind spot that had persisted for over a century.
Andrew Holmes and Olena Bukalo's work at the NIH, together with Lindsay Halladay's contributions from the University of Arizona, establishes that the brain's emotional processing architecture is a collaboration between cell types, not a neuronal monologue. Fear memories don't just live in neural circuits. They are maintained by a partnership between neurons that fire the signals and astrocytes that decide whether those signals persist or fade.
For the millions of people living with PTSD, anxiety disorders, and phobias, this partnership offers something concrete: a new therapeutic target. The road from mouse models to clinical treatments is long and uncertain, but it now begins from a fundamentally different starting point. We spent a century trying to fix fear by talking exclusively to neurons. The cells that were actually holding the fear in place were the ones we thought were just holding the brain together.
Sources
- These overlooked brain cells may control fear and PTSD - ScienceDaily
- Astrocytes Discovered as Architects of Fear Memory - Neuroscience News
- Astrocytes enable amygdala neural representations supporting memory - Nature
- Astrocytes coordinate fear memories alongside neurons - PNAS Journal Club