The African trypanosome has a problem. It lives in the human bloodstream, which is one of the most hostile environments on earth for anything that isn't a blood cell. The immune system constantly patrols for invaders, tagging unfamiliar proteins for destruction. To survive, the parasite that causes sleeping sickness wraps itself in a molecular disguise, a dense coat of proteins that looks innocuous to immune cells. When the immune system finally learns to recognize one disguise, the parasite switches to another. It has been doing this for millions of years, and until recently, nobody understood exactly how it managed the trick with such precision.
Now a team of researchers led by Dr. Joana Faria at the University of York has answered a question that parasitologists have been asking for four decades: how does the trypanosome keep its protein disguise so perfectly calibrated? The answer, published in Nature Microbiology, turns out to involve a protein that works like a molecular shredder, sitting inside the parasite's gene expression machinery and destroying specific genetic messages the instant they're produced. The parasite doesn't just control what it makes. It controls what it destroys.
The Invisibility Cloak
To understand why the shredder matters, you need to understand the cloak. African trypanosomes, the single-celled parasites that cause sleeping sickness (also called African trypanosomiasis), survive in the bloodstream by coating their entire surface with a single type of protein called variant surface glycoprotein, or VSG. Each parasite wears approximately 10 million copies of the same VSG molecule, forming a dense shield so uniform that the immune system initially treats it as background noise.
The trick is in the switching. The trypanosome genome contains roughly 2,000 different VSG genes, but only one is active at any time. When the immune system finally produces antibodies against the current VSG coat, a small fraction of parasites have already switched to a different version. Those survivors repopulate the bloodstream wearing a new disguise, and the cycle begins again. This process, called antigenic variation, is why sleeping sickness can persist for years and why no vaccine has ever been developed against it. The target keeps changing before the immune system can lock on.
The gene that produces the active VSG sits in a specialized location called the expression site, alongside several helper genes called Expression Site Associated Genes (ESAGs). These helpers perform support functions: transporting nutrients, helping the parasite interact with host cells, and facilitating aspects of immune evasion. The entire package, the VSG gene and its associated helpers, gets transcribed together as one long stretch of genetic instructions.
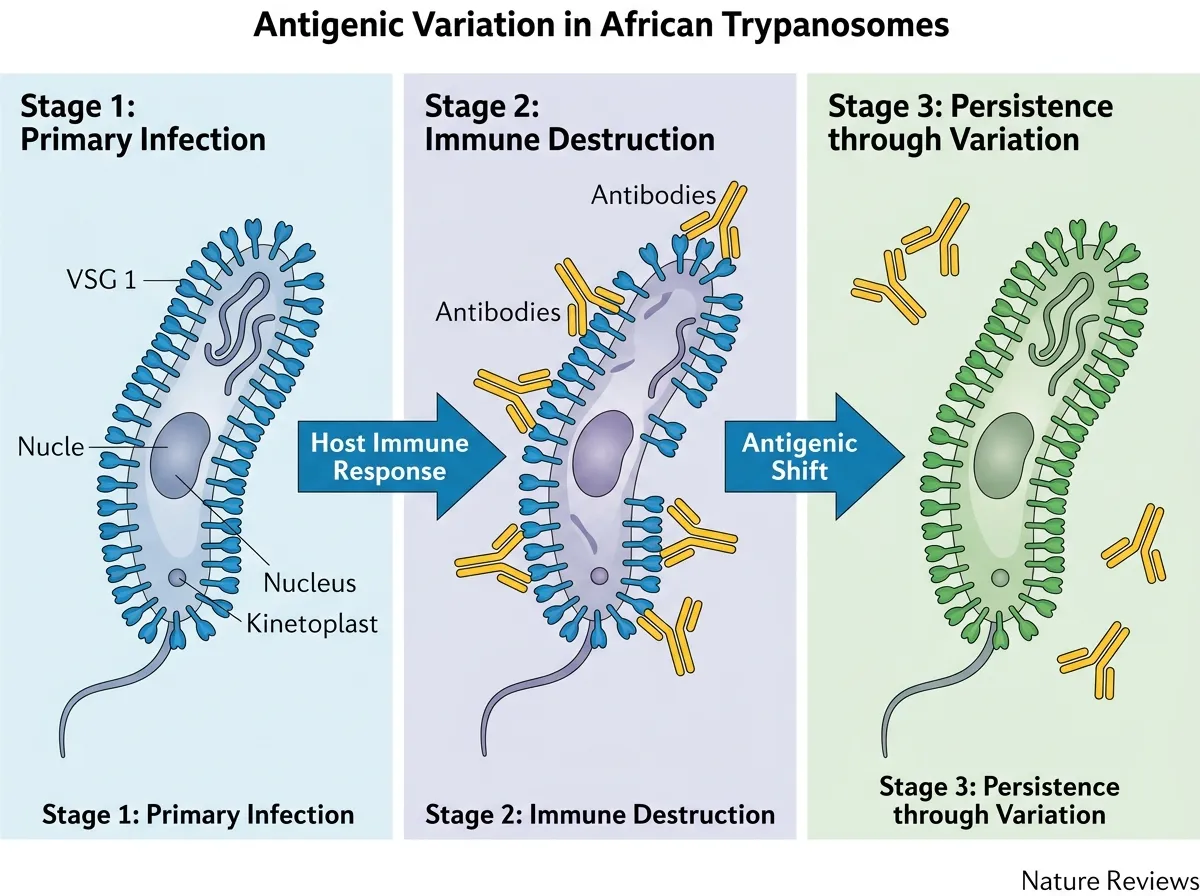
The Puzzle That Lasted 40 Years
Here's where the mystery comes in. When scientists studied gene expression at the trypanosome's active expression site, they expected to find roughly equal amounts of each protein produced. After all, the VSG gene and the ESAG helper genes sit on the same stretch of DNA and get transcribed together as a single polycistronic message. In most organisms, genes transcribed together are produced in similar quantities.
But that's not what happens. The trypanosome produces massive amounts of VSG protein, enough to coat its entire surface, while producing only tiny quantities of the helper proteins. The ratio is wildly lopsided: the VSG output dwarfs the ESAG output by orders of magnitude. For 40 years, researchers knew this imbalance existed but couldn't explain the mechanism behind it. Something was selectively reducing the ESAG messages after transcription, but nobody could identify what or how.
The question mattered beyond basic curiosity. Understanding how the parasite controls its surface coat is directly relevant to developing treatments. If you can disrupt the mechanism that maintains the cloak, you could potentially expose the parasite to the immune system. But you can't disrupt a mechanism you haven't identified, and until 2026, the identity of this one remained unknown.
The Molecular Shredder
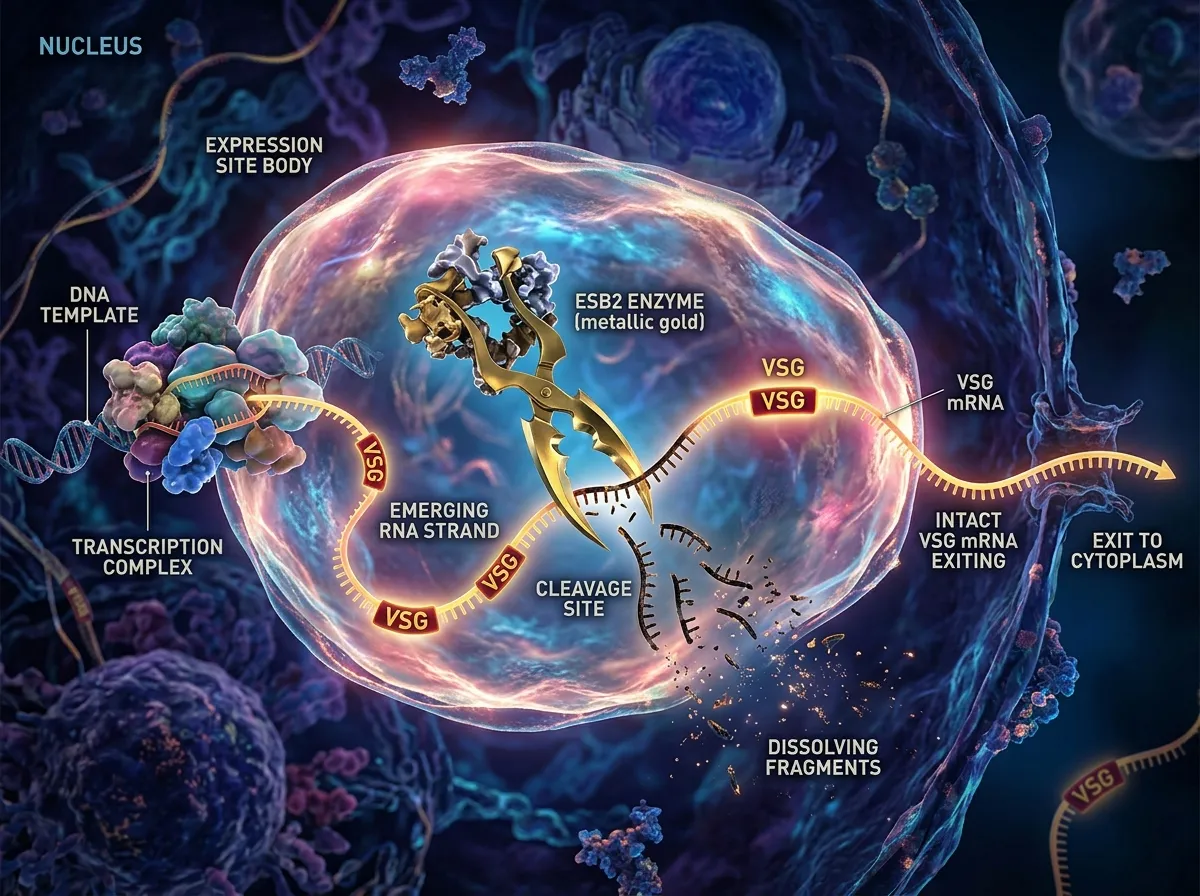
Dr. Faria's team found the answer by looking inside a structure called the Expression Site Body (ESB), a specialized compartment within the parasite's nucleus where the active expression site is transcribed. Using a combination of protein identification techniques and genetic manipulation, the researchers discovered a previously uncharacterized protein sitting inside the ESB. They named it ESB2.
ESB2 functions as an RNA endonuclease, an enzyme that cuts RNA molecules at specific points. But what makes ESB2 remarkable is its selectivity and its location. It sits directly inside the transcription factory, positioned to intercept genetic messages the moment they're produced. When the polycistronic message containing both VSG and ESAG instructions rolls off the transcription machinery, ESB2 immediately shreds the ESAG portions while leaving the VSG instructions intact.
The result is elegant in its brutality. The parasite transcribes everything together, then destroys the parts it doesn't want in bulk, letting the VSG instructions survive to be translated into the millions of protein copies that form the cloak. Rather than using the complex regulatory systems that most organisms employ to fine-tune gene expression, the trypanosome takes a simpler approach: make everything, then selectively destroy what you don't need. It's the biological equivalent of printing an entire newspaper and then shredding every section except the front page.
When the researchers knocked out ESB2, the carefully maintained imbalance collapsed. ESAG protein levels rose dramatically, and the VSG coat became compromised. The parasite lost its ability to maintain its disguise at the concentration needed to evade immune detection. The shredder, it turned out, wasn't just a fine-tuning mechanism. It was essential for survival.
Why Destruction Beats Regulation
The ESB2 mechanism reveals something counterintuitive about biological strategy. Most organisms regulate gene expression by controlling how much of each gene gets transcribed in the first place, adjusting the rate of production like a factory controlling which assembly lines are running. The trypanosome does something conceptually different: it runs every assembly line at full speed and then destroys the products it doesn't want at the end.
This approach might seem wasteful, and in one sense it is. The parasite expends energy transcribing ESAG genes only to shred the resulting RNA immediately. But from an evolutionary perspective, the strategy has a specific advantage. Antigenic variation requires the parasite to switch its active expression site periodically, swapping from one VSG gene to another. If the regulatory machinery were built into the transcription process itself, every switch would require reconfiguring that machinery. By using a post-transcriptional shredder instead, the parasite can switch expression sites without changing anything about how it regulates protein ratios. ESB2 simply shreds the helper messages at every expression site equally, regardless of which one is active.
This kind of solution, where destruction is used instead of regulation, has parallels elsewhere in biology. The immune system itself uses controlled protein destruction to manage inflammation, and certain cancer cells evade treatment by degrading the very receptors that drugs target. What's unusual about the trypanosome is the precision. ESB2 doesn't degrade RNA randomly. It targets specific transcripts within a specific cellular compartment, acting more like a programmed editor than a blunt instrument.
A Disease That Still Kills
The scientific elegance of the ESB2 mechanism shouldn't obscure the human stakes. Sleeping sickness, caused by Trypanosoma brucei transmitted through tsetse fly bites, has plagued sub-Saharan Africa for centuries. The disease progresses in two stages: an initial phase of fever, headaches, and joint pain, followed by a neurological phase where the parasite crosses into the brain, causing confusion, disrupted sleep cycles (hence the name), and eventually death if untreated.
Thanks to sustained public health efforts, the number of reported cases has dropped dramatically, from nearly 40,000 per year in the late 1990s to fewer than 1,000 annual cases in recent years. But the disease remains endemic in 36 countries across sub-Saharan Africa, and other pathogen-driven diseases continue to reveal unexpected biological mechanisms that complicate treatment. Current drugs for sleeping sickness are often toxic, difficult to administer, and limited in availability. The neurological stage remains especially challenging to treat because most drugs cannot cross the blood-brain barrier.
Identifying ESB2 opens a potential new avenue for intervention. If a drug could inhibit ESB2's shredding activity, the parasite's coat regulation would collapse, exposing it to immune attack. The parasite would essentially lose its invisibility cloak. This remains a long way from clinical reality: drug development timelines typically span a decade or more, and the parasite's ability to evolve resistance is well-documented. But the discovery gives researchers a target they didn't have before, which is how drug development begins.
The Deeper Question
The ESB2 discovery is a resolution, but it also opens new lines of inquiry that the York team and collaborators from six countries are already pursuing. How did this system evolve? The trypanosome lineage is ancient, diverging from other eukaryotes roughly a billion years ago. The ESB2 mechanism may represent a strategy that predates the more familiar gene regulation systems found in animals and plants, or it may be a convergent solution that evolved independently under the intense selective pressure of living inside a vertebrate immune system.
There's also the question of whether similar mechanisms exist in related parasites. Trypanosoma cruzi, which causes Chagas disease in the Americas, and various Leishmania species use different immune evasion strategies but face the same fundamental challenge: hiding from a host that is actively trying to destroy them. Whether post-transcriptional RNA destruction plays a role in those organisms remains unexplored.
What the York team has demonstrated, at minimum, is that one of the most successful survival strategies in biology operates on a principle that seems almost paradoxical: the parasite's power lies not in what it builds, but in what it chooses to destroy. For 40 years, scientists looked for a mechanism that turned genes on or off. The answer turned out to be a molecular shredder that was running the entire time, sitting in the factory, cutting the messages that the parasite couldn't afford to let through.
Sources
- Specialized RNA decay fine-tunes monogenic antigen expression in Trypanosoma brucei - Nature Microbiology
- Scientists solve 40-year-old biological mystery behind sleeping sickness - Phys.org
- University of York scientists solve 40-year-old biological mystery behind Sleeping Sickness - University of York
- Parasitic sleeping sickness creates 'invisibility cloak' to hide in humans for years - Popular Science
- Scientists discover "molecular shredder" that helps deadly parasite evade the immune system - ScienceDaily